Cross-sectional study
- Study Type
- Design
- Measures/Example
- Study Type
- Observational
- Design
- Collects data from a group of people to assess frequency of disease (and related risk factors) at a particular point in time.
- Asks, “What is happening?””
- Measures/Example
- Disease prevalence.
- Can show risk factor association with disease, but does not establish causality.
Case-control study
- Study Type
- Design
- Measures/Example
- Study Type
- Observational and retrospective
- Design
- Compares a group of people with disease to a group without disease.
- Looks for prior exposure or risk factor.
- Asks, “What happened?”
- Measures/Examples
- Odds ratio (OR).
- “Patients with COPD had higher odds of a history of smoking than those without COPD had.”
Cohort study
- Study Type
- Design
- Measures/Example
- Study Type
- Observational and prospective or retrospective
- Design
- Compares a group with a given exposure or risk factor to a group without such exposure.
- Looks to see if exposure increased the likelihood of disease.
- Can be prospective (asks, “Who will develop disease?”) or retrospective (asks, “Who developed the disease [exposed vs. nonexposed]?”).
- Measures/Example
- Relative risk (RR).
- “Smokers had a higher risk of developing COPD than nonsmokers had.”
Twin concordance study
- Design
- Measures/Example
- Design
- Compares the frequency with which both monozygotic twins or both dizygotic twins develop same disease.
- Measures/Example
- Measures heritability and influence of environmental factors (“nature vs. nurture”).
Adoption study
- Design
- Measures/Example
- Design
- Compares siblings raised by biological vs. adoptive parents.
- Measures/Example
- Measures heritability and influence of environmental factors.
Clinical trial
- Experimental study involving humans.
- Compares therapeutic benefits of 2 or more treatments, or of treatment and placebo.
- Study quality improves when study is randomized, controlled, and double-blinded (i.e., neither patient nor doctor knows whether the patient is in the treatment or control group).
- Triple-blind refers to the additional blinding of the researchers analyzing the data.
Drug Trials: Phase I
- Typical Study Sample
- Purpose
- Typical Study Sample
- Small number of healthy volunteers.
- Purpose
- “Is it safe?”
- Assesses safety, toxicity, and pharmacokinetics.
Drug Trials: Phase II
- Typical Study Sample
- Purpose
- Typical Study Sample
- Small number of patients with disease of interest.
- Purpose
- “Does it work?”
- Assesses treatment efficacy, optimal dosing, and adverse effects.
Drug Trials: Phase III
- Typical Study Sample
- Purpose
- Typical Study Sample
- Large number of patients randomly assigned either to the treatment under investigation or to the best available treatment (or placebo).
- Purpose
- “Is it as good or better?”
- Compares the new treatment to the current standard of care.
Drug Trials: Phase IV
- Typical Study Sample
- Purpose
- Typical Study Sample
- Postmarketing surveillance trial of patients after approval.
- Purpose
- “Can it stay?”
- Detects rare or long-term adverse effects.
- Can result in a drug being withdrawn from market.
Evaluation of diagnostic tests
- Uses 2 × 2 table comparing test results with the actual presence of disease.
- TP = true positive
- FP = false positive
- TN = true negative
- FN = false negative
- Sensitivity and specificity are fixed properties of a test (vs. PPV and NPV).

Sensitivity (true-positive rate)
- Definition
- Equations
- Definition
- Proportion of all people with disease who test positive, or the probability that a test detects disease when disease is present.
- Value approaching 100% is desirable for ruling out disease and indicates a low false-negative rate.
- High sensitivity test used for screening in diseases with low prevalence.
- Equations
- = TP / (TP + FN)
- = 1 – false-negative rate
- If sensitivity is 100%
- TP / (TP + FN) = 1
- FN = 0
- All negatives must be TNs
- SN-N-OUT = highly SeNsitive test, when Negative, rules OUT disease
Specificity (true-negative rate)
- Definition
- Equations
- Definition
- Proportion of all people without disease who test negative, or the probability that a test indicates non-disease when disease is absent.
- Value approaching 100% is desirable for ruling in disease and indicates a low false-positive rate.
- High specificity test used for confirmation after a positive screening test.
- Equations
- = TN / (TN + FP)
- = 1 – false-positive rate
- If specificity is 100%
- TN / (TN + FP) = 1
- FP = 0
- All positives must be TPs
- SP-P-IN = highly SPecific test, when Positive, rules IN disease
Positive predictive value (PPV)
- Definition
- Equation
- Definition
- Proportion of positive test results that are true positive.
- Probability that person actually has the disease given a positive test result.
- PPV varies directly with prevalence or pretest probability
- High pretest probability –> high PPV
- Equation
- = TP / (TP + FP)
Negative predictive value (NPV) (51)
- Definition
- Proportion of negative test results that are true negative.
- Probability that person actually is disease free given a negative test result.
- NPV varies inversely with prevalence or pretest probability
- High pretest probability –> low NPV
- Equation
- = TN / (FN + TN)

Incidence vs. prevalence
- Equations
- Comparison
- Equations
- Incidence rate = # of new cases in a specified time period / Population at risk during same time period
- Incidence looks at new cases (incidents).
- Prevalence = # of existing cases / Population at risk
- Prevalence looks at all current cases.
- Incidence rate = # of new cases in a specified time period / Population at risk during same time period
- Comparison
- Prevalence ≈ incidence rate × average disease duration.
- Prevalence > incidence for chronic diseases (e.g., diabetes).
- Incidence and prevalence for common cold are very similar since disease duration is short.
Odds ratio (OR)
- Definition
- Equations
- Definition
- Typically used in case-control studies.
- Odds that the group with the disease (cases) was exposed to a risk factor (a/c) divided by the odds that the group without the disease (controls) was exposed (b/d).
- Equations
- OR = (a/c) / (b/d) = ad / bc

Relative risk (RR)
- Definition
- Equations
- Definition
- Typically used in cohort studies.
- Risk of developing disease in the exposed group divided by risk in the unexposed group
- e.g., if 21% of smokers develop lung cancer vs. 1% of nonsmokers, RR = 21/1 = 21
- If prevalence is low, RR ≈ OR.
- Equations
- RR = [a / (a+b)] / [c / (c+d)]

Relative risk reduction (RRR)
- Definition
- Equations
- Definition
- The proportion of risk reduction attributable to the intervention as compared to a control.
- e.g., if 2% of patients who receive a flu shot develop flu, while 8% of unvaccinated patients develop the flu, then RR = 2/8 = 0.25, and RRR = 1 – RR = 0.75
- Equations
- RRR = 1 – RR
Attributable risk (AR)
- Definition
- Equations
- Definition
- The difference in risk between exposed and unexposed groups, or the proportion of disease occurrences that are attributable to the exposure
- e.g., if risk of lung cancer in smokers is 21% and risk in nonsmokers is 1%, then 20% (or .20) of the 21% risk of lung cancer in smokers is attributable to smoking.
- Equations
- AR = [a / (a+b)] - [c / (c+d)]

Absolute risk reduction (ARR)
- Definition
- Equations
- Definition
- The difference in risk (not the proportion) attributable to the intervention as compared to a control
- e.g., if 8% of people who receive a placebo vaccine develop flu vs. 2% of people who receive a flu vaccine, then ARR = 8% - 2% = 6% = .06.
- Equations
- ARR = [c / (c+d)] - [a / (a+b)]

Number needed to treat
- Definition
- Equation
- Definition
- Number of patients who need to be treated for 1 patient to benefit.
- Equation
- NNT = 1/ARR.
Number needed to harm
- Definition
- Equation
- Definition
- Number of patients who need to be exposed to a risk factor for 1 patient to be harmed.
- Equation
- NNH = 1/AR.
Precision
- The consistency and reproducibility of a test (reliability).
- The absence of random variation in a test.
- Random error—reduces precision in a test.
- Increased precision –> decreased standard deviation.
Accuracy
- The trueness of test measurements (validity).
- The absence of systematic error or bias in a test.
- Systematic error—reduces accuracy in a test.

Selection bias
- Definition
- Examples
- Berkson bias
- Loss to follow-up
- Healthy worker and volunteer biases
- Strategies to reduce bias
- Definition
- Nonrandom assignment to participate in a study group.
- Most commonly a sampling bias.
- Examples
- Berkson bias
- A study looking only at inpatients
- Loss to follow-up
- Studying a disease with early mortality
- Healthy worker and volunteer biases
- Study populations are healthier than the general population
- Berkson bias
- Strategies to reduce bias
- Randomization
- Ensure the choice of the right comparison/reference group
Recall bias
- Definition
- Example
- Strategy to reduce bias
- Definition
- Awareness of disorder alters recall by subjects
- Common in retrospective studies.
- Example
- Patients with disease recall exposure after learning of similar cases
- Strategy to reduce bias
- Decrease time from exposure to follow-up
Measurement bias
- Definition
- Example
- Strategy to reduce bias
- Definition
- Information is gathered in a way that distorts it.
- Example
- Hawthorne effect — groups who know they’re being studied behave differently than they would otherwise
- Strategy to reduce bias
- Use of placebo control groups with blinding to reduce influence of participants and researchers on experimental procedures and interpretation of outcomes
Procedure bias
- Definition
- Example
- Strategy to reduce bias
- Definition
- Subjects in different groups are not treated the same.
- Example
- Patients in treatment group spend more time in highly specialized hospital units
- Strategy to reduce bias
- Use of placebo control groups with blinding to reduce influence of participants and researchers on experimental procedures and interpretation of outcomes
Observer-expectancy bias
- Definition
- Example
- Strategy to reduce bias
- Definition
- Researcher’s belief in the efficacy of a treatment changes the outcome of that treatment
- aka Pygmalion effect; self-fulfilling prophecy
- Example
- If observer expects treatment group to show signs of recovery, then he is more likely to document positive outcomes
- Strategy to reduce bias
- Use of placebo control groups with blinding to reduce influence of participants and researchers on experimental procedures and interpretation of outcomes
Confounding bias
- Definition
- Example
- Strategies to reduce bias
- Definition
- When a factor is related to both the exposure and outcome, but not on the causal pathway
- Factor distorts or confuses effect of exposure on outcome
- Example
- Pulmonary disease is more common in coal workers than the general population
- However, people who work in coal mines also smoke more frequently than the general population
- Strategies to reduce bias
- Multiple/repeated studies
- Crossover studies (subjects act as their own controls)
- Matching (patients with similar characteristics in both treatment and control groups)
Lead-time bias
- Definition
- Example
- Strategy to reduce bias
- Definition
- Early detection is confused with increased survival
- Seen with improved screening techniques.
- Example
- Early detection makes it seem as though survival has increased, but the natural history of the disease has not changed
- Strategy to reduce bias
- Measure “back-end” survival (adjust survival according to the severity of disease at the time of diagnosis)
Measures of central tendency
- Mean
- Median
- Mode
- Mean = (sum of values)/(total number of values).
- Median = middle value of a list of data sorted from least to greatest.
- If there is an even number of values, the median will be the average of the middle two values.
- Mode = most common value.
Measures of dispersion
- Standard deviation
- Standard error of the mean
- Standard deviation = how much variability exists from the mean in a set of values.
- Standard error of the mean = an estimation of how much variability exists between the sample mean and the true population mean.
- σ = SD, n = sample size
- SEM = σ / sqrt(n)
- SEM decreases as n increases
Normal distribution
- Gaussian, also called bell-shaped.
- Mean = median = mode.

Bimodal distribution
- Suggests two different populations
- e.g., metabolic polymorphism such as fast vs. slow acetylators; suicide rate by age

Positive skew
- Typically, mean > median > mode.
- Asymmetry with longer tail on right.
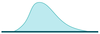
Negative skew
- Typically, mean < median < mode.
- Asymmetry with longer tail on left.

Null Hypothesis (H0)
- Hypothesis of no difference
- e.g., there is no association between the disease and the risk factor in the population

Alternative Hypothesis (H1)
- Hypothesis of some difference
- e.g., there is some association between the disease and the risk factor in the population

Table: Power, Type 1 Error, Type 2 Error, and Correct
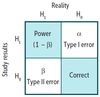
Correct result
- Stating that there is an effect or difference when one exists
- Null hypothesis rejected in favor of alternative hypothesis
- Stating that there is not an effect or difference when none exists
- Null hypothesis not rejected
Type I error (α)
- Definition
- α & p
- Definition
- Also known as false-positive error
- Stating that there is an effect or difference when none exists
- Null hypothesis incorrectly rejected in favor of alternative hypothesis
- α = you saw a difference that did not exist (e.g., convicting an innocent man).
- α & p
- α is the probability of making a type I error.
- p is judged against a preset a level of significance (usually < .05).
- If p < 0.05, then there is less than a 5% chance that the data will show something that is not really there.
Type II error (β)
- Definition
- β & power
- Definition
- Also known as false-negative error.
- Stating that there is not an effect or difference when one exists
- Null hypothesis is not rejected when it is in fact false
- β = you were blind to a difference that did exist (e.g., setting a guilty man free).
- β & power
- β is the probability of making a type II error.
- β is related to statistical power (1 – β), which is the probability of rejecting the null hypothesis when it is false.
- Increase power and decrease β by:
- Increasing sample size
- There is power in numbers.
- Increasing expected effect size
- Increasing precision of measurement
- Increasing sample size
Meta-analysis
- Pools data and integrates results from several similar studies to reach an overall conclusion.
- Increase statistical power.
- Limited by quality of individual studies or bias in study selection.
Confidence interval
- Definition
- Equation
- 95% & 99% CI
- If the 95% CI for a mean difference between 2 variables includes 0
- If the 95% CI for odds ratio or relative risk includes 1
- If the CIs between 2 groups do not overlap
- If the CIs between 2 groups overlap
- Definition
- Range of values in which a specified probability of the means of repeated samples would be expected to fall.
- Equation
- CI = range from [mean – Z(SEM)] to [mean + Z(SEM)].
- 95% & 99% CI
- For the 95% CI, Z = 1.96.
- The 95% CI (corresponding to p = .05) is often used.
- For the 99% CI, Z = 2.58.
- For the 95% CI, Z = 1.96.
- If the 95% CI for a mean difference between 2 variables includes 0
- Then there is no significant difference and H0 is not rejected.
- If the 95% CI for odds ratio or relative risk includes 1
- H0 is not rejected.
- If the CIs between 2 groups do not overlap
- Significant difference exists.
- If the CIs between 2 groups overlap
- Usually no significant difference exists.
t-test
- Checks differences between means of 2 groups.
- Tea is meant for 2
- Example: comparing the mean blood pressure between men and women.
ANOVA
- Checks differences between means of 3 or more groups.
- 3 words: ANalysis Of VAriance
- Example: comparing the mean blood pressure between members of 3 different ethnic groups.
Chi-square (χ²)
- Checks difference between 2 or more percentages or proportions of categorical outcomes (not mean values).
- Pronounce Chi-tegorical
- Example: comparing the percentage of members of 3 different ethnic groups who have essential hypertension.
Pearson correlation coefficient (r)
- Definition
- Positive vs. negative r value
- Coefficient of determination
- Definition
- r is always between -1 and +1.
- The closer the absolute value of r is to 1, the stronger the linear correlation between the 2 variables.
- Positive vs. negative r value
- Positive r value –> positive correlation.
- Negative r value –> negative correlation.
- Coefficient of determination = r2 (value that is usually reported).
Disease Prevention
- Primary
- Secondary
- Tertiary
- Quaternary
-
Primary
- Prevent disease occurrence (e.g., HPV vaccination).
-
Secondary
- Screening early for disease (e.g., Pap smear)
-
Tertiary
- Treatment to reduce disability from disease (e.g., chemotherapy)
-
Quaternary
- Identifying patients at risk of unneccessary treatment, protecting from the harm of new interventions
Medicare and Medicaid
- Both
- Medicare
- Medicaid
- Both
- Federal programs that originated from amendments to the Social Security Act.
- Medicare
- Available to patients ≥ 65 years old, < 65 with certain disabilities, and those with end-stage renal disease.
- MedicarE is for Elderly
- Medicaid
- Joint federal and state health assistance for people with very low income.
- MedicaiD is for Destitute
