Some uses of SPECTROSCOPY:
Mixture of compounds -> Chromatography -> single compound
Single Compound -> KNOWLEDGE OF STRUCTURE
Bio System + Compound –> Compound Localization / MoA / Protein-protein Interactions
What is SPECTROSCOPY?
Study of matter through its interaction w/ diff compounds of the EM spectrum,
interaction of energy with matter
understand how LIGHT interacts with MATTER
how to use it to learn about variety of properties in a molecule
4 First (smallest) Wavelength Regions
& what they measure
Gamma rays - n/a
X-Rays - core/inner level electrons
UV & Visible** = **valence / outer electrons
3 LAST (largest) Wavelength Regions
& what they measure
IR** **- molecular VIBRATIONS
Microwave - molecular ROTATIONS
Radio - NUCLEAR SPIN
What does a spectrum measure?
how the sample MODIFIES THE LIGHT interacting with it
- Light source (EM wave) -> Sample
- Change in Light
- Change in Sample
- =photochemistry
- leads to DEGRADATION, typically irreversible
- =photochemistry
Typical process of “small molecule” structure ELUCIDATION
Compound ->
IR / UV-Vis / MS ->
1D NMR / 2DNMR ->
IDENTIFIED STRUCTURE
X-Ray Cytallography SKIPS all those steps
X-Ray Crystallography
Energy Interaction / Central Concept
DIFFRACTION = bending of light around an object
- x-ray beam -> crystal of a SINGLE PURE crystal compound
- bend/defract/scatter light in 3D space, 360 rotation
- onto a detector, which is then computed
- bend/defract/scatter light in 3D space, 360 rotation
X-Ray Crystallography
Used for / Works on?
ribosome / how proteins fit together / antibiotic mechanism
has to be a PURE / SINGLE crystal
(has a HUGE number of PRECISELY ordered, IDENTICAL molecules)
Performed on both:
SMALL or LARGE
molecules 100-2k Da or >2k Da
X-Ray Crystallography
strengths & weaknesses
- Strength:
- Possible to determine entire structure** including **relative or absolute stereochemistry of small molecules
Good if you can get it to crystallize in the first place
- Weakness:
- Crystallization process is laborious; certain classes of molecules are difficult/nearly impossible to crystallize
•Crystallization is difficult to make
IR (infrared) Spectroscopy
Energy interaction / Central Concept
IR spectroscopy measure
MOLECULAR VIBRATIONS
- X = Wave number (cm-1)
- Y = % Transmittance, similar to absorbance but FLIPPED
- Specific functional groups act in a
- VERY SPECIFIC & CONSISTANT WAY
- made a reference sheet for comparison
- VERY SPECIFIC & CONSISTANT WAY
IR (infrared) Spectroscopy
Used for / Works For?
presence of FUNCTIONAL GROUPS
Bond Distance
Natural product elucidation, but NMR/MASS spectroscopy is better
need to compare to a reference / cheat sheet
IR (infrared) Spectroscopy
Strength / Weakness
- Strength:
- Indicates potential presence of FUNCTIONAL GROUPS
- including bond distance
- Indicates potential presence of FUNCTIONAL GROUPS
- Weakness:
- does NOT indicate CONNECTIVITY of functional groups
- ex. we don’t know how 8 carbons + 10 hydrogens are bound,
- dont know the actual molecule STRUCTUR
- ex. we don’t know how 8 carbons + 10 hydrogens are bound,
- does NOT indicate CONNECTIVITY of functional groups
Atomic Absorption (aa) Spectroscopy
Energy Interaction / Central Concept
EMISSION SPECTRA of each element produces a unique fingerprint
Flame Test - similar to FP (Flame Photometry)
Flame converts metal ions -> atoms
measures Light Intensity
AA Spectroscopy
Works on / Common uses
Determination of METALS (Hg / Pb / etc)
from LIQUID** **(urine / plasma / serum)
AA Spectroscopy
Strengths / Weaknesses
- Strength:
- __used for Metals in Liquids/Solution
- Weakness:
- Low throughput, requires manual operation
- _DESTRUCTIVE_
- Low throughput, requires manual operation
UV-Vis Spectroscopy
Energy Interaction / Central Concept
- Spec of UV & Vis regions measures valence / outer electrons
- Molecules absorb EM radiation
- as atoms pass from ground state -> Excited State
- Molecules absorb EM radiation
- More effectively a molecule ABSORBS LIGHT (given wavelength,
- –> GREATER the extent of light ABSORBANCE
- views CHROMOPHORE
- functional group that absorbs UV radiation
Chromophore
Functional group that absorbs UV Radiation
views CONJUGATION
organic molecules between 190-800 nm
detected by UV / PDA Detectors
Name the Process of rapidly identifying compounds that are
KNOWN CHEMICAL SCAFFOLDS
DEREPLICATION
used in UV/PDA
UV / PDA Detectors
PhotoDiode Array
both view Light Absorption** of **Chromaphores
Photodiode Array (PDA) Detectors = pretty much the same as UV detection but with ALL WAVELENGTHS
IF DETECTABLE BY UV IT IS ALSO DETECTABLE BY PDA,
but not the other way around
UV / PDA Spectroscopy
S / W
- Strengths:
- UV fingerprint may indicate the CLASS of a molecul
- Weakness:
- does NOT distinguish between ISOMERS within a molecular class
- if the structural change does not involve the CHROMOPHORE
- Need an EXTENSIVE UV fingerprint
- some classes do not absorb in UV regionn
- does NOT distinguish between ISOMERS within a molecular class
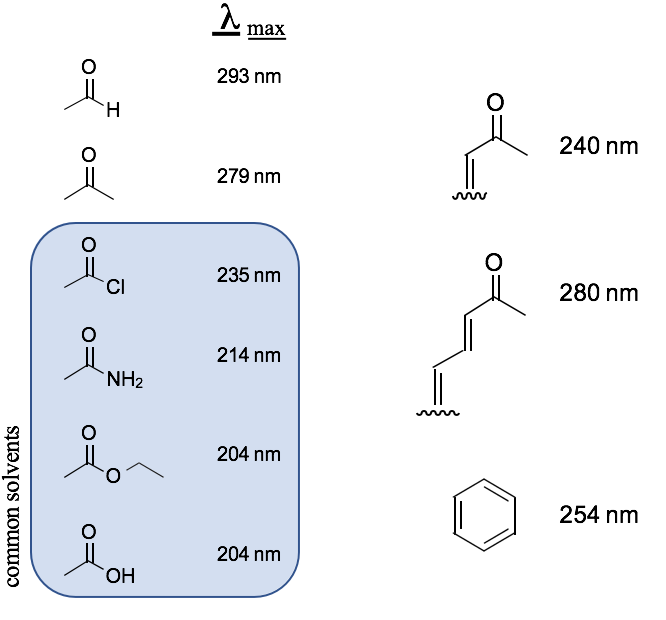
Common Chromophores
Blue box = solvent interference
chromophore wavelength OVERLAPS
w/ wavelength of solvent
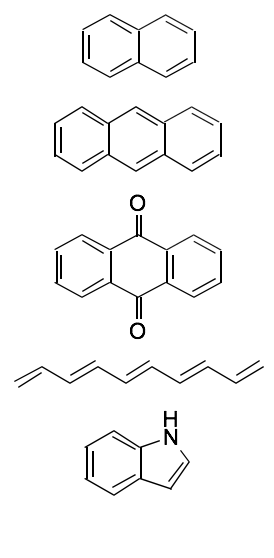
Chromophres with more complex conjugation
Complex conjugation = more DISTINCT UV Fingerprint
UV / PDA Spectroscopy
Works on / Common uses
UV fingerprint is helpful for INDENTIFYING compound class if:
MOST of the structure contributes to the UV Chromophore
but we can link a chromophore to a reactive functional group
to make an UV-Active material
ELSD
Evaporative Light Scattering Dector
Concept / Detects for?
Replacement or used in TANDEM w/ UV/PDA Detector
does not rely on spectroscopic qualities of molecule
only detects a PHYSICAL PRESENCE
of a compound, either SENSES or NOT,
needs physical blockage to be detected


