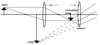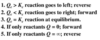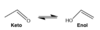Chem-Phys Flashcards
Do cell membranes made with phosphatidyl-choline/-ethanolamine require a counter ion? why or why not?
No. they are charge balanced b/c (+) charge of choline/ethanolamine group offsets (-) charge of phosphate grp
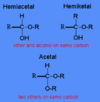
“Difference between isomers…
- structural
- geometrical
- conformational
- optical”
- structural: different connectivity
- geometrical: cis vs. trans.
- conformational: interchangable structures by rotation abount a bond
- optical: enantiomers—»same connectivity, different spatial orientation about chiral center”

treatment of Oleic acid with D2/Pd would yield a compound with how many chiral centers?

what factors increase membrane rigidity?
1) inc. degree of saturation
2) inc. # of carbons
which of the following alcohols are oxidized by Na2Cr2O7?
- phenols
- methyl/1º alcohols
- 2º alcohols
- 3º alcohols
methyl, primary, & secondary alcohols
How can you quickly identify whether a change in oxidation state is an oxidation or reduction? (i.e. S–S —» S–C)
If an atom is bound to a more electronegative atom —» oxidation
If an atom is bound to a less electronegative atom —» Reduction
what is the difference in structure of an enamine vs. imine

What forms when an aldehyde is treated with a grinard reagent?
a 2º alcohol is formed
Pinacol Rearrangement
Pinacol Rearrangement: converts a vicinal diol (OH groups on neighboring carbons) into a ketone via an alkyl shift.

of the 4 types of compounds, which are/are not reduced by NaBH4?
- phenol
- 3º alc
- ketone
- ester
- phenol —» no
- 3º alc —» no
- ketone —» yes
- ester —» no


if a food molecule (i.e. carb, fat, protein, etc.) isn’t metabolized by the digestive system, how many dietary calories are contributed to the consumer?
0 Cal are contributed to the consumer b/c anything that isn’t metabolized cannot provide any energy & ∴ no calories.
why is a solution of NaNO2(aq) basic?v
NO2- reacts w/H2O to form OH- ions, thereby inc. pH of solution
if there are 60 million nuclei of a radioactive atom (half-life of 430 years), how many years would it take before there are 3.75E^6 nuclei remaining?
- 1 half-life = [(# nuclei) / 2]
- (3.75E6)/(60E^6) = 1/16 = [4 half-lifes]
- 4(430) = 1720
What are 2 different units for electric field?
- Newton/Coulomb (N/C)
- Volt/Meter (V/m)
– 1(V) = 1 (J/C)
what type of interference occurs when two waves are out of phase by 180º?
Destructive Interference: occurs when two waves that meet when they are half a wave difference out of phase (180º)
how does the period of a waveform change as you move up through the 1st, 2nd, & 3rd harmonic? what about wavelength? frequency?
- the period (T) DEC. as you move up a harmonic level (i.e. 1st—»2nd)
- the wavelength DEC. as harmonic level increases
- if 1st harmonic = [100 Hz], 2nd = [200 Hz], 3rd = [300 Hz], etc.
what is the only effect that the photon frequency has on ejected electrons?
- photon frequency only affects the ejected e-‘s KE —» ∴ inc. in frequency = inc. in KE = inc. speed of ejected e-
- photon energy = cathode work function + KEelectron
if someone drinks an extremely hypertonic/hypersaline solution, why would this person be at risk of death?
ingested salt would be absorbed into the blood, creating a hypertonic environment outside the cells of the body—»causes water to flow out of the cell causing it to shrivel
How does adding salt to water affect it’s boiling point & vapor pressure?
1) adding salt inc. BP of water
2) adding salt dec. VP of water—»∴ as solute inc., the rate of water molecules breaking the liquid surface dec.
•BP—»the temperature at which VP of solution = VP of the atmosphere
what allows H2O to exhibit the unique property where it’s solid state (ice) is less dense then the liquid (water)
• the bent structure of H20 (bond angle ~104.45º) & the high degree of hydrogen bonding yields a hexagonal crystalline structure w/alot of space between molecules
1) Β- Decay
2) What is the product of I-131 B- decay?
1) conversion of neutron to proton & loss of 1e-
• element undergoing B-decay will convert to an element w/1 additional H+
2) Xe-131
What are the 4 main types of radioactive decay?
1) α-Decay: emission of alpha particle (2 protons & 2 neutrons [2+])
2) B- / B+ Decay:
- B- = neutron converted to H+ and B- particle (e-) ejected
- B+ = H+ converted to neutron and B+ particle (positron) emitted
3) Gamma Decay: emission of gamma ray (high energy photon) and release of ionizing radiation
4) Electron Capture: nucleus “grabs” an e- —» converts H+ to a neutron
if a child and an adult are exposed to the same contamination levels of I-131, would the child, compared with the adult, recieve a higher, lower, or equal relative dose?
- B/c children weigh less, the same quantity of external I-131 results in a higher concentration of the isotope in the body
- higher concentration = inc. likelyhood of DNA damage & cancer
If 2-methylundecanal and 2-methylundecanoic acid are separated by distillation, which compound would be found in the collecting flask and which would be found in the initial boiling flask?
• distillation separates compounds based on difference in BP w/the lower BP molecule evaporating off first ∴ …
-Collection Flask = 2-methylundecanal
-Boiling Flask = 2-methylundecanoic acid
What is the formula for the velocity of an object dropped from a given hieght (h)?
v = √2gh
what is one way to quickly identify whether or not a carbon is a stereogenic center?
for a carbon to be a tetrahedral stereocenter, it must be bonded to 4 different substituents
if a runner undergoes a turning acceleration to make a turn, how would you determine the magnitude of force experienced by the foot due to the ground?
- Fnet = √(Fnormal^2) + (Fturn^2)
- Fturn is ⊥ to Fnormal ∴ resultant = Fnet
For a polyprotic acid, what are the requirements for a H+ to produce a visible inflection point?
- a H+ will only produce a visible inflection point if the H is relatively strong (low pKa)
- pka1 vs. pka2 must differ by at least a factor of 10^4
1) what is the equation for the moles of acid of a monoprotic acid at the equivalence point?
2) If you are given the titration curve for captopril w/NaOH & the concentration of NaOH, how would you determine the mols of captopril in the original analyte solution
1) Mb x Vb = Ma x Va = moles acid
2) determine volume of NaOH at the inflection pt of the titration curve (middle of steep portions) and use the concentration to determine the number of moles of NaOH at the inflection point. Mol OH = Mol Captopril
Newton’s Laws
1) Newton’s 1st Law: states thaat within a reference frame, an object remains at rest or at a constant velocity unless an external force acts upon it
• Fnet = 0 ; at equillibrium
2) Newton’s 2nd Law: Fnet = ma
3) Newton’s 3rd Law: when body A exerts a force on body B, body B exerts and equal and opposite force on body A
• Fa on b = -Fb on a
How would you determine whether a data point with error bars is significant?
if error bars overlap between data points that are being compared, then the difference between those pts. is NOT SIGNIFICANT
Within the ETC, are e- passed from species w/less positive to more positive reduction potential or vice versa?
- within the ETC, e- are passed from species w/less positive reduction potential to those w/more positive reduction potential
- O2 = final e- acceptor—»∴ possess most postive standard reduction potential
standard reduction potential (Eº)
- tendency for a species to spontaneously become reduced
- defined relative to standard hydrogen electrode (2H+(aq) + 2e-(aq) —» H2(g))
- Units: volts
- (+) Eº —» stubstance ‘wants’ to be reduced
- (–) Eº —» substance not prone to reduction
- Eº Cell = Eºcathode + Eºanode [total standard potential generated by a galvanic cell]
For a redox reaction, where are specieces reduced vs. oxidized?
1) Cathode (+) —» electrode where reduction occurs
2) Anode (–) —» electrode where oxidation occurs
If you’re given the energy released by NADH thru the ETC (at 50% efficiency), how would you determine if energy produced by the rxn would be sufficient for the generation of 3 ATP molecules.
- energy released by NADH = -52.5 kcal/mol
- at 50% efficiency, only 26 kcal/mol could be used to produce ATP
- 3 ATP —» 1 ATP (7.3 kcal/mol) x 3 = 22 kcal/mol ∴ enough energy would be produced to generate 3 ATP from the e-pair from NADH
Refractive index
- refractive index (n) helps reference how a waves speed will change within different mediums releative to it’s speed in a vaccum
- n = c/v
(v=velocity through medium, c=speed of light)
Snell’s Law
- relates how the propagation speed of a wave changes from one medium to another —» relates the refractive index to how much the light bends upon entering the new mediuim
- n1sin(Ø1) = n2sin(Ø2)
What is the hybridization of XeF4?
sp3d2
What is the relationship between ∆Hf & the rxn rate?
- ∆Hf (heat of formation) = energy required for the formation of a molecule
- lower ∆Hf = quicker rxn rate
what is relative stabilization energy (RSE)? how does RSE relate to the stabilizing effects of a molecule?
RSE —» (–) RSE = greater stabilization

Hess’ Law
∆Hrxn = ∑∆Hf [products] - ∑∆Hf [reactants]
What is the difference between enthalpy vs. entropy?
Enthalpy = heat energy in a system
Entropy = energy in a closed system that is unavailable to do work
What is the relationship between electronegativity (w/respect to the substituent) and its stabilizing effects on a carbocatrion?
- carbocations are unstable b/c of high concentration of positive charge ∴ substituents that contribute e- density to the carbocation will charge balance the carbocation
- EWGs pull e- densityt away from carbocation, further decreasing its stability
1) Gibbs Free Energy Equation
2) is it possible for an endothermic rxn to proceed spontaneously?
1) ∆G = ∆H - T∆S
2) YES. If ∆S is a large, (+) number, then it could make the overall ∆G of a rxn (–)
spontaneity relationship table for ∆H, ∆S, and

What does the power of a study refer to?
Power: refers to the ability of a study design to detect a real statistically significant effect, and it is primarily affected by the size of the study and the size of the effect - larger sample = more power
Capacitor
• Capacitance (C): C = εA/d
- A: area of plates
- d: distance btwn plates
• Medical applications of capacitors (i.e. defibrillators to store charge and electrical PE)
? atm = ? mmHg = ? Pa
1 atm = 760 mmHg = 10^5 Pa
Homotropic regulation (HR)
HR: when a molecule serves as a substrate for it’s target enzyme, as well as a regulatory molecule of the enzyme’s activity
- O2 = homotropic allosteric modulator of Hb
- HR is what causes Hb graph to have sigmoidal shape
why is hydrostatic pressure (Hp) for liquids linear, but not for gases?
- Hp for liquids remains linear b/c the density of the fluid doesnt change at different depths
- the density of gases vary depending on the forces applied to them—»gases are compressible while liquids are not
deposition
- phase change from gas to solid
- exothermic
if an indicator dyte is needed for a titration of acetic acid w/NaOH, what should the pH of the indicator be?
- b/c acetic acid = weak acid, and NaOH = strong base, the pH at the titration endpoint would be expected to be greater than 7
equation to calculate unknown concentration or volume for a titration
NaVa = NbVb
- N: normality (mol/L)—»molarity must be converted to normality for polyprotic acids/polyvalent bases
If a person is excercising against a 50-W load for 5 min, what is the total work performed by the muscle?
- P=Wt
- W = P/t = (50J/s)/(300s) = 150 kJ
- what equation allows you to determine the percent remaining for a decaying isotope if the number of half lives is given? 2. if you are told that a sample of 2000g must be replaced when the mass decays to 250, given the info from question (1) how would you detemine the number of half lives to reach that point?
- (0.5^n) = % remaining [n: # 1/2 lives]
- 250/2000 = 1/8 (1/8) = (1/2)^n
Formula for Mechanical Advantage of an inclined plane
MA = hypotenuse/height
If each coupling step for a 10AA peptide produced a 95% yield, what would be the final yield for the peptide?
(0.95)^9 = 0.63 ≈ 60%
**only raised to a power of 9 b/c the cycle would only be run 9 times for a 10 AA peptide
the flow rate of a heart valve is 4E^-4 (m^3/s). what speed would result within a blood vessel w/a cross sectional area of 5E^-6 (m^2)?
**UNITS**
- Q = Av
- v = Q/A
- (4E^-4) / (5E^-6) = 8E^-2 (m/s)
Pyruvate Dehydrogenase Complex
- Converts pyruvate—»acetyl CoA -generates 1 NADH
- complex of 3 enxymes
a) pyruvate dehydrogenase
b) dihydrolipoyl transacetylase
c) dihydrolipyl dehydrogenase - Requires 5 coenzymes:
a) TPP b) FAD c) NAD d) CoA e) lipolate
Surfactant
- amphipathic molecules (hydrophilic head and hydrophobic tail)
- surfactant adsorbs to air-water-alveoli interface—»reducing surface tension and total force resisting expansion
• elastic recoil force of the airway and surface tension of water lining the airway oppose expansion of alveoli—»surfactant reduces the force needed to expand alveoli
Inspiration
- contraction of diaphram & external intercostal muscles—»results in dec. in intrapleural pressure (neg. presure)
- Neg. presure (rel. to Patm at entry to airway) causes airflow through respiratory tract to end at alveoli
What is the equation for the gibbs free energy change of a phase change from liquid to solid?
∆G = G(l) - G(s)
∆G = ∆H -T∆S
- ∆H = enthalpy of formation
- ∆S = entropy of formation
Quantum Numbers/Electron Configuration
- principle quantum number (n): energy level of the e- •higher n = higher energy
- Angular (azimuthal) momentum # (l): subshell of n •l = n–1 • s=0, p=1, d=2, f=3
- Magnetic quantom # (ml): •ml = (-l), 0, (+l)
- Spin quantum number (ms): spin orientation •ms = +/- 1/2
equation to calculate the energy of an electron’s n level
En = -Rh/(n^2) Rh
(Rydberg Constant) = different for each element
what is range in wavelength for the visible spectrum of electromagnetic signals? what color does each end correspond to?
- Range = [400nm - 700nm]
- 400nm = purple (high energy)
- 700nm = red (low energy)
if isotpe X & Y both undergo B- decay, what is the expected ratio of total atomic mass of material X vs. material Y?
- half life X = 5 years
- half life Y = 1 year
- Ratio = 1:1
•the atomic mass of an atom doesnt change when B- decay takes place b/c the transition from neutron to proton has a negligible change in atomic mass
If an electron were ejected from an sp, sp2, or sp3 porbital, which of these would emit electromagnetic radiation w/the shortest wavelength?
sp orbital—»shorter wavelength emissions correspond to higher energy electromagnetic emmissions ∴ b/c it would require the most energy to eject an electron from an sp orbital, it would have the shortest wavelength
Hertz
Hz, Frequency s^-1
Newton
N, Force, kg m/s^2
Pascal
Pa, Pressure, N/m^2
Joule
J, Work/Energy, Nm
Newton-meter
Nm, Torque
Watt
W, Power, J/s
Coulomb
C, Charge
Volt
V, Electric potential, J/C
Farad
F, Capacitance, C/V
Ohm
(Omega), Resistance, V/A
Weber
You don’t need this, seriously
Tesla
T, Magnetic field
Y32 is a genetically encoded fluorescent biosensor, and the spectral ratio of peaks of excitation spectrum are a direct representation of ATP-ADP ratio. If its proposed that differences in ATP-ADP ratios are due to variations in Y32 expression, would the student be correct?
- NO—» Y32 fluorescent intensity ratio of peaks is a reflection of its ATP-ADP ratio
- use of ratio = intrinsic normalization—»∴ doesnt matter how many biomolecules are in the sample, the ratio provides normalization that makes the values reliable
Whats the most likely cause of a shift in the emission spectra of a fluorescent molecule where wavelength increases?
an inc. in wavelength corresponds to a dec. in energy —» loss of energy occurs when e- returns to ground state b/c it was excited w/the same amount of energy that would have produced the first emission spectrum .
if a fluorescent molecule is pH-sensitive, why might this cause problems when using it to monitor changes in the energy balance of a cell?
pH will change over time in a cell, and changes to emission spectrum might be reflection of the change in pH and not whatever it is supposed to be indicating
If log(MW) = n, what is the molecular weight of that molecule equal to?
MW = 10^n [Da]
If the pKa of a weak acid = 3.5, what would be the most accurate guess for the pH of a [.1M] solution? -1.0, 2.3, 3.5, or 4.1?
- 2.3
- if it were a strong acid then pH = 1
- if [HA] = [A-] then pH = pKa = 3.5
Calculating Galvonic cell potential
- galvanic cell = spontaneous rxn 2. cell potentials (Eº) always > 0 [i.e.(+)] 3. if given the reduction potential for both components of the galvonic cell—»reverse the sign of the less positive reduction potential (i.e. oxidation potential) •then simply add the ox/red potentials directly —» Eº = Eºr + Eºo
Ohm’s Law
V = IR
Capacitance equation & Units
- C = Q/V = κε0A/d
- k = dialectric constant
- [κ ∝ C] —» Qκ = 4Q0 when dialectric constant is added to a capacitor
- ε0 = constant (not needed for MCAT)
- A = area of the plates
- d = distance between plates
- k = dialectric constant
- Capacitantce must always be (+)
- Unit: farads (F)
- q: coulomb (C)
- V: volt (V)

Phosphorus Acid Molecular Formula
H3PO3
Difference between ‘-ous’ acids vs. ‘-ic’ acids?
- “-ous”: acids have 1 fewer O atam than “-ic” counterpart acid
- Ex: Phosphorous Acid (H3PO3) vs. Phosphoric Acid (H3PO4)
Ksp
- Ksp (solubility product) = [A-]^2[B^2+]
- (A-) & (B2+) = two ions of aq ionic compound
- ion concentration raised to the power of its stoichiometric coefficient
- High Ksp=more soluble | Low Ksp = less soluble
If a mechanical leg mimics a spring’s compression, and it absorbs 125J of KE after compressing 10cm, whats the spring constant?
- leg absorbing KE and converts to elastic potential enerfy
- KE = 1/2kx^2 • k = 2KE/x^2 | k = 25,000 N/m
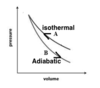
What is the relationship of volume and temperature for an ideal gas? (P is kept constant)
- direct relationship (when pressure is constant)
- V1/T1 = V2/T2
• T must be in Kelvin (K)
What are the units required for temperature whenever dealing with an ideal gas?
Kelvin




On what basis does gas-liquid chromatograph separate compounds?
Vaporizes the compounds. The first peaks exhibit the lowest molecular weight and intermolecular forces.
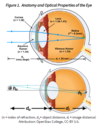
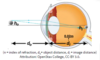
What is the thin lens formula?
The strength of the eye lens is given as the inverse of the focal length of the eye lens. 1/O + 1/I = S . O stands for the distance someone can see objects. I is the lens to retina distance.
How do you calculate the index of refraction?
It is equal to the ratio of the speed of light in a vacuum / the speed of light in the medium
What is intensity of radiation proportional to?
The number of photons emitted
What is the difference between threonine and serine?
Threonine has a secondary alcohol while serine has a primary alcohol
What is the hybridization of an octahedral compound?
sp3d2
What bond does glycogen synthase catalyze?
The a-1,4-glycosidic linkage of an individual glucose molecule to the main chain of glycogen
How can you use the thin lens equation to find the height of an image?
1/o + 1/i = 1/f . You can calculate the distance (i) of an image if you know F and O. The height of an image is the ratio of i/o
o: object distance
i: image distance
f: focal length
What is +∆S?
An increase in entropy and more disorder as if 2 reactants became 3 products
What will occur if a homogenous catalyst cannot be separated from the products at the end of a reaction?
The reaction will still occur, but the products will be contaminated with the catalyst
Name the strong acids:
HCl, HNO3, HI, HClO4, HClO3
Name the strong bases:
KOH, NaOH, Ba(OH)2, CsOH, Sr(OH)2, Ca(OH)2, LiOH, RbOH
Express the amount of light energy that is converted to other forms between the flouescence excitation and emission events in J/photon:
E=hc/(1/ye-1/yf)
How do you calculate Kcat?
Kcat = Vmax/[E]
Absorption of UV light by organic molecules always results in what process?
Excitation of bound electrons
What is the half life of radioactive material?
The time it takes for half of all the radioactive nuclei to decay into their daughter nuclei
Which phosphate is used by kinases in phosphoryl transfer?
gamma-Phosphate
How is cooperativity expressed in a graph?
A sigmoidal curve
What phenomenon causes static air to be drawn into the mask when oxygen flows?
Venturi effect
Why do G-C residues have higher boiling points than A-T residues?
G-C bonds exhibit a higher π-stacking strength
What are the difference between phospholipids and storage lipids?
Phospholipids consist of a glycerol molecule with two fatty acid tails, and a phosphate group modified by an alcohol. Storage lipids have three fatty acid tails ester linked to a single glycerol
What are Lewis bases?
Lone pair donors
What is a coordinate covalent bond?
A lewis acid-base interaction between a metal cation and an electron pair donor
What will HCl react with first: ammonia or water?
NH3 because it is a much stronger base
What is the coordination number?
The number of molecules that coordinate covalent bond to a central metal cation
What is henderson hasselbach formula for acids and what is 10^-1?
pH=pKa+log[base/acid] ; 10^-1=.1
What is the solubility product constant expression for CaCO3? What is the concentration of a saturated solution of CaCO3 if the Ksp=4.9*10^-9?
Ksp=[Ca][CO3] ; if the Ksp is 4.9*10^-9 then [Ca] is the sqrt of that: 7*10^-5
What is the difference in sound intensities between 20 and 40dB?
A difference of 20dB is 2 factors of 10 so 100 times more intense
How is energy converted in a battery that has a resistor?
Chemical to electrical to thermal energy
What is the bond formed by protein secondary structure?
Hydrogen bonds between the backbone amide protons and carbonyl oxygens
What is the magnification formula and what would the di be for an object 20cm to the left of a lens?
M = -di/do . to the left of the lens the image has a negative value