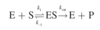Chapter 2. Enzymes Flashcards
(72 cards)
Fill in the blank.
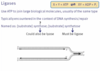
The molecules that are acted on by enzymes are called ____.
substrates
What are ribozymes?
Biological catalysts composed of RNA instead of peptides.
What is the activation energy (Ea) of a reaction?
The input of energy needed to overcome the barriers in the reaction.
Finish the sentence.
Enzymes decrease the activation energy, so molecular collisions ____.
occur more often
What are the six classes of enzymes?
- Oxidoreductases
- Transferases
- Hydrolases
- Lyases
- Isomerases
- Ligases
What are oxidoreductases?
- Class of enzymes that catalyze redox reactions.
- Transfer electrons between biological molecules.

When looking at oxidoreductases, the ____ is the electron donor and the ____ is the electron acceptor.
reductant; oxidant
Fill in the blank.
Oxidoreductases often have a cofactor that acts as an ____.
electron carrier
Oxidoreductases often have a cofactor that acts as an electron carrier. Give an example of three cofactors associated with oxidoreductases.
NAD+, NADP+, and heme
Which class of enzyme catalyzes the movement of a functional group from one molecule to another?
Transferases

Fill in the blank.
Transferases often employ ____.
coenzyme donors
Transferases often employ coenzyme donors. Give an example of one common coenzyme donor associated with transferases.
Coenzyme A
Fill in the blank.
Kinases and polymerases are classified as a ____ enzyme.
transferase
What are kinases?
- Transferase enzyme.
- Catalyze the transfer of a phosphate group, generally from ATP, to another molecule.
What is a polymerase?
- Transferase enzyme.
- Catalyze the transfer of nucleotides.
Which class of enzyme catalyzes the breaking of a compound into two molecules using the addition of water?
Hydrolases

What is the reverse reaction of hydrolases?
Dehydration synthesis
What is a phosphatase?
- Hydrolases enzyme.
- Cleaves a phosphate group from another molecule.
Which type of enzyme catalyzes the cleavage of a single molecule into two products?
Lyases
Which group of enzymes often forms rings or multiple bonds to reform octets?
Lyases
Fill in the blanks.
Lyases require ____ phosphate group(s) to cleave molecules, and ____ phosphate group(s) to bring molecules together.
one; two
Which class of enzyme catalyzes the rearrangement of bonds within a molecule?
Isomerases

Fill in the blanks.
Isomerases interconvert between ____ and ____.
constitutional isomers and stereoisomers
Which class of enzyme catalyzes addition or synthesis reactions, generally between large similar molecules, and often require ATP?
Ligases