Wk 3 In-Class Nichols Flashcards
(19 cards)
What is this?

- Amyloidosis, which tends to be “hyaline” (smooth and featureless) & can be confluent (late)
- How can you tell the difference between this and fibrosis? Congo red or immunostain for amyloid P
A 68-year-old white male bartender in Memphis, TN, with a history of smoking presents in February with worsening dyspnea and cough over several days. What is your differential dx?
- Should include:
1. Heart failure due to ischemic heart disease (perhaps a silent MI)
2. Pneumonia: bacterial or viral
3. Pulmonary embolism
4. Other autoimmune, idiopathic diseases we haven’t learned about
This myocardium has 3 zones: delineate, describe and diagnose each.

- Zone 1: hypereosinophilia + loss of nucleus = coagulative necrosis
- Zone 2: vacuolated (cleared) cytoplasm = myocytolysis
- Zone 3: interstitial edema and viable myocytes
What is the difference between interstitial and confluent fibrosis?
- Interstitial (patchy) fibrosis: between individual cells or groups of cells
- Confluent is bigger than that, usually because some of the myocytes have died and gone away
What are re-entrant cardiac arrhythmias?
-
Re-entry: common mechanism by which altered impulse conduction leads to tachyarrhythmias
1. In such rhythm, electrical impulse circulates repeatedly around a reentry path, recurrently depolarizing a region of cardiac tissue - In normal cardiac conduction, e/electrical impulse that originates in SA node travels in an orderly, sequential fashion through rest of heart, ultimately depolarizing all the myocardial fibers
1. Refractory period of each cell prevents immediate reexcitation from adjacent depolarized cells, so impulse stops when all of heart muscle has been excited
2. Conduction blocks that prevent rapid depolarization of parts of myocardium can create an envo conducive to continued impulse propagation and reentry
If the banker who had a heart attack on the lawn’s mother had witnessed him collapsing, called 911 and paramedics resuscitated him in time for coronary angioplasty a few hours later, reperfusion of his myocardial infarction would alter it by causing less what?
Neutrophil infiltration
A 68-y/o white male bartender in Memphis, TN, with a hx of smoking presents in February with worsening dyspnea and cough over several days. Dyspnea is on exertion and he has chronic cough. He awakes at night sweaty, chilly, short of breath and coughing, but he has long had repeated nighttime awakening. He feels sick, has a stuffy nose, sore throat, muscle aches and joint aches, but his answers to questions are all vague and he does not know what day it is.
He appears ill and is obese. His temperature is 38, heart rate 110, blood pressure 110/60, respirations 29 and saturation 93%. His heart has an irregular rhythm, but no murmurs. He has bilateral basal pulmonary crackles. His abdomen is mildly distended, but not tender. He has mild ankle edema. His skin is cool and moist. What is your differential diagnosis?
- Should include causes of atrial fibrillation
1. Heart failure can cause atrial fibrillation, but so can pneumonia or pulmonary embolism
2. He has fever, which suggests infection
3. He has ankle edema and bibasilar crackles, which suggests heart failure
4. Cool, moist skin suggests shock
What is this?

- Collagen - it is usually wavy in the interstitium
- No nuclei, and less than the size of an RBC (7 microns)
Below are the labs of a patient that comes in with worsening dyspnea (on exertion) and cough over several days, sweaty, chilly, awakening at night, stuffy nose, sore throat, confusion, low BP, high HR, pulmonary crackles, and an irregular rhythm.
BNP is 400 (normal
What is your diagnosis?
- Elevated BNP confirms a diagnosis of heart failure
- Elevated WBC count and neutrophilia goes with a diagnosis of infectious pneumonia (could be flu and superimposed pneumococcal or staph pneumonia)
- Pt has heart failure due to combo of underlying undiagnosed ischemic heart disease and influenza pneumonia w/sepsis that precipitated heart failure
- Vasodilation + INC metabolism of sepsis cause INC HR and cardiac output to meet INC demand -> if coronary atherosclerosis limits blood flow to the heart, the demand on the pump can cause it to fail
- The patient’s heart failure was partly septic and partly cardiogenic
What is the classic unreperfused microscopic pathology of an MI?
- Thin wavy myocytes, first visible as early as 1/2 hour, if present, = earliest finding
- Coagulation necrosis = hypereosinophilia + loss of striations + nuclear pyknosis, then karyorrhexis, karyolysis, loss, first visible at 4-12 hours
- Neutrophilic infiltration (acute inflammation) follows myocyte necrosis, first visible at 6-12 hours
What are the reperfusion effects in an MI?
- Smaller than it would have been
- More patchy than it would have been
- More contraction band necrosis
- More macros
- More interstitial fibrosis
- Hemorrhage into it
- Accelerated inflammation and repair
- Diffusion of inflammation and repair
- Fewer neutrophils (bc cytokine release altered)
This 37-year-old white female with a 22-year history of insulin-dependent diabetes was hospitalized 10 hours prior to death with chest pain and shortness of breath. Three months before, she had begun to experience progressive weakness and for the previous 3 weeks, she had increasing dyspnea on exertion and worsening of a chronic cough. Can you describe the gross pathology, and then make two diagnoses?

- Left ventricle shows concentric hypertrophy with wall thickness well over the upper limit of normal (1.4 cm) and numerous small, irregularly shaped patches of white scarring
- The left ventricular hypertrophy most likely represents hypertensive heart disease
- The scars most likely represent old myocardial infarction
Does warm skin always differentiate sepsis from other types?
No - only sometimes (don’t count on it)
Why is a patchy, old MI like this the substrate for malignant ventricular arrhythmias?
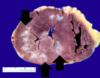
- Each little, irregularly shaped scar is a wall of non-conducting tissue that an electrical signal can “bounce off of”
- If it “bounces off” more than one or just one at the wrong angle, that can send it backward or sideways to myocytes that pass it on before the normal signal, creating a malignant re-entrant ventricular tachycardia
- Re-entrant VT from scarring is typically MONOMORPHIC, or has unvarying shape of the QRS, correlated with originating from a single site
55-y/o black male banker with a BMI of 31 and 48 pack-year hx of smoking ending 17 years ago has a 15-year history of diabetes mellitus treated with metformin, hypercholesterolemia treated with atorvastatin, and hypertension treated with amlodipine and benazepril. He volunteers to mow his mother’s lawn, collapses while doing so and is found dead. Autopsy shows the attached image of his heart -> cause of the patient’s death was?

Myocardial infarction less than 4 hours old
What do you see?

- Thin, wavy myocytes - in this case, all dead for awhile because their nuclei have faded away
- Notice how these are all in register (unlike collagen)
68-year-old white male bartender in Memphis, TN, with a history of smoking presents in February with worsening dyspnea and cough over several days. On further questioning, the dyspnea is on exertion and he has a chronic cough. He awakes at night sweaty, chilly, short of breath and coughing, but he has long had repeated nighttime awakening. He feels sick, has a stuffy nose, sore throat, muscle aches and joint aches, but his answers to questions are all vague and he does not know what day it is. What is your differential diagnosis?
- Should broaden to include:
1. Shock because the brain window of his body shows malfunction
2. Feeling ill = malaise, and together with nasal congestion, sore throat, muscle aches and a cough, this should suggest influenza
What should you think when you see the words, “irregular rhythm?”
Re-entry or fibrillation
What does it mean if the BNP is up?
Heart failure (can help distinguish between this and PE)


