L13. Pain Flashcards
(43 cards)
What is the definition of pain?
An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage
A subjective experience that can be experienced independently of tissue damage
What are the 4 different classes of pain? Categorise them into 2 major types
Adaptive and Protective
- Nociceptive pain
- Inflammatory pain
Maladaptive and pathological
- Neuropathic pain
- Functional pain syndromes
Pain is divided into four distinct stages. Name and describe these stages?
- Transduction = detection of noxious stimuli
- Transmission = electrical signals through afferent neurons through the peripheral and then the central nervous systems
- Perception = processing of the information into sensory and discriminative and emotional and aversive responses
- Modulation = we infer that there is some kind of descending pathway that modifies the ascending pathway in some way because nociception doesn’t always lead to pain.

What are some examples of noxious stimuli that are sensed in the transduction stage?
Heat
Acid
Noxious Cold
Pressure
Chemicals
Tactile
All dected through different types of fibre
You can only percieve pain if you are conscious. What can be inferred by this fact about the pain pathway?
That sensory detection of pain (nociception) can occur independently of the sensation of pain
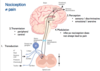
Describe the afferent pathway of the pain information (electrical information) from the nociceptor to the cortex
- Nociceptors in the skin detect the stimuli and relay information through the afferent neuron to the dorsal horn of the spinal cord
- Two tracts carry the information up to the lateral thalamus and limbic centres
- These then relay information up to the cortex for processing of emotional/aversive and sensory/discriminatory information.
Describe nociceptor neurons
They are specialised afferent sensory nerve cells that respond to high levels of stimuli (high threshold).
They have peripheral bodies, cell body neurons (DRG) and central processes to the spinal cord.
Stimulation by noxious stimuli to a nociceptor sensory neuron will illicit what rmajor esponses? [3]
- Pain sensation
- Autonomic responses (like sweating, flight or fight, etc)
- The withdrawal reflex

What is the main evolutionary reason for nociception?
Nociception is an adaptive, high threshold pain receptor that gives an early warning system (protection)
Eg. Some people have defects in Na channels in these nociceptive neurons and thus are unable to conduct information through them. These people constantly suffer damaged tissues due to inability to experience pain.
Describe the nerve fibres of nociceptors (what are they called)
- They are free nerve endings
- They have fine axons
- Have no specialised transducers
- They respond/fire to their stimuli with more intensity the longer the stimuli is applied (sometimes long after it is gone)
c-fibres
aδ-fibres

What is the difference between the two nociception fibres: Aδ fibres and the C fibres?
C fibres are unmyelinated and are very thin (<1.5um). This means that they conduct information quite slowly (< 3m/s)
Aδ fibres are myelinated and are also thin (1.5-4um). They conduct information with medium-fast speed (3-30m/s - which is slower than touch but faster than C-fibres)
Through what afferent system will migraines and tooth and jaw pains transmit through?
The trigeminal afferent system (through the trigeminal ganglion)
What types of pain do the dorsal root afferent fibres relay?
Somatic (skin, muscular and bone)
and
Visceral (non-somatic organs)
Nociceptors have high thresholds, what is meant by this?
They only fire in response to potentially damaging stimuli. It takes a lot of force or stimulus to activate an action potential within them
(eg. heat nociceptors only fire at temperatures >43 degrees which is damaging to the skin).

What is meant by first and second pain in terms of nociception?
Because of the different speeds of conduction of the Aδ fibres (faster) and the C fibres (slower) there is a resultant separation in the two information sets.
The brain will interpret the first pain as sharp and localised folled by a later pain that is dull and throbbing.

C fibres continue to fibre once the external stimulus has gone away (tonically fire), why would they do this?
Especially if there has been damage to the tissue.

Different tissue have different mixtures and ratios of the receptors. Describe the distribution of the fibres in response to noxious heat in the hand (hairy vs. glabrous)
Hairy skin (the back of the hand) has both a type II Aδ and C-fibres meaning that the hand feels both a sharp and a slow, burning feeling in response to heat.
Glabrous skin (palm) only has the C-fibres and thus will not experience the sharp pain before the burning kicks in.

What part of the dorsal horn do nociceptors predominatly project to?
Predominantly to the peripheral layers of the dorsal horn (particularly to laminae I-V)
C-fibres to laminae I-III
Aδ fibres to laminae IV and V
The C fibres are never deeper than the Aδ fibres
(note that Ab fibres are for mechanoreceptors)

At what level does pain decussate?
At the same level the input arrived in the spinal cord (segmental)

After the nociceptor axons enter the spinal cord, what happens to them from there?
Nociceptive dorsal afferents enter the dorsal horn and then synapse with second order neurons (in the peripheral the dorsal laminae) and these second order neurons decussate to the ventral horn and send axons up and down the spinal cord through the spinothalamic/anteriorlateral tract.
Describe the major difference between the path of mechanoreception axon fibres and nociceptor fibres in the brainstem
They both enter at the dorsal horn but the nociceptor fibres synapse early and decussate while the mechanoreceptors don’t have the major synapse and move ipsilaterally through the dorsal columns.

Describe the spino-nociceptive reflex
This is the ability to move the limb as a defensive reflex (withdrawal) can occur purely through a spinal reflex (no higher input).
Thus noxious nociceptive stimuli can drive a circuit that will move the limb (protective reflex) without involvement of the brain or brainstem
NOCICEPTION CAN OCCUR INDEPENDENTLY OF PAIN

What are the two tracts that carry nociceptive information to the brain from the periphery? What is the major difference between them?
- The Spino-brachial tract: carries information from Ad and C fibres to the limbic system which relays info to the frontal cortex for emotional and aversive responses
- The Lateral spino-thalamic tract: carries information from Ab fibres to the lateral thalamus which relays to the frontal cortex for sensory and discriminatory information about the pain.
What is the difference between nociceptive pain and inflammatory pain?
Inflammatory pain is nociceptive pain that has worsened or is different as a result of tissue damage.













