Pharmacology-Chemotherapy Flashcards
(67 cards)
Melphalan/chlorambucil
Class III Alkylating agents “Nitrogen mustards”.
Cyclophosphamide
Class III Alkylating agent “Nitrogen mustard”. Requires hepatic activation. Different side effects that melphalan and chlorambucil: hepatotoxicity, other nitrogen mustard side effects are eliminated.
Lomustine (CCNU)
Class III Alkylating agent. Chloroethylnitrosurea. Good for treating brain tumors due to lipophilicity. Delayed bone marrow suppression.
Carmustine (BCNU)
Class III Alkylating agent. Chloroethylnitrosurea. Has 3 chloroethyl groups for cross-linking of DNA. Good for treating brain tumors due to lipophilicity. Delayed bone marrow suppression.
Cisplatin
Alkylating agent. Platinum cross links DNA. Used in testicular, ovarian and head and neck cancer
Oxaliplatin
Alkylating agent. Platinum cross links DNA. Used for colorectal cancer
Carboplatin
Alkylating agent. Platinum cross links DNA. Less renal toxicity than cisplatin and oxaliplatin
Procarbazine
Alkylating agent. Nonspecific alkylator after hepatic activation. Used for Hodgkin’s lymphoma.
Vincristine/vinblastine
Class II “Natural product”. Blocks polymerization of microtubules and prevents mitosis. Side effect of peripheral neuropathy due to signal transduction interruption from microtubule inhibition.
Paclitaxel
“Natural product”. Freezes microtubules in polymerized state and prevents mitosis.
Docetaxel
“Natural product”. Synthetic analogue of paclitaxel.
Etoposide/Teniposide
“Natural product”. Inhibits DNA topoisomerase II, prevents DNA replication due to supercoiling. Used for Hodgkin’s lymphoma, diffuse histiocytic lymphoma and small cell lung cancer.
Irinotecan
“Natural product”. Inhibits DNA topoisomerase I, prevents DNA replication due to supercoiling. Used for metastatic colorectal cancer.
Topotecan
“Natural product”. Inhibits DNA topoisomerase I, prevents DNA replication due to supercoiling. Used for ovarian and small cell lung cancer.
Doxorubicin/Daunorubicin
“Natural product”. Intercalates into DNA and prevents replication. Generates free radicals that chemically react with DNA. Inhibits DNA topoisomerase.
Bleomycin
“Natural product”. After DNA binding domain links up with DNA, Fe-O2 arm swings in, breaks up pyrimidine ring and causes single-stranded breaks.
Methotrexate
Antimetabolite. Inhibits dihydrofolate reductase, preventing FH2 -> FH4. This prevents synthesis of dTMP and thus prevents DNA synthesis. Good for use in combination with 5-fluorouracil.

Leucovorin
FH4 used for rescue in patients treated with high-dose methotrexate that have their bone marrow nearly wiped out.
5-fluorouracil
Antimetabolite. Irreversibly inhibits thymidylate synthase, preventing conversion of dUMP to dTMP and thus prevents DNA synthesis. Good for use in combination with methotrexate.

Cytosine arabinoside
Antimetabolite. Inhibits DNA polymerases and prevents elongation of 3’ end if incorporated into DNA where cytosine is normally incorporated, causes premature termination.
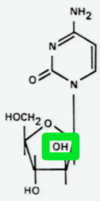
Gemcitabine
Antimetabolite. Inhibits DNA polymerases and prevents elongation of 3’ end if incorporated into DNA where cytosine is normally incorporated, causes premature termination, like Ara C. Good activity in advanced breast cancer, non-small cell lung cancer, pancreatic carcinoma and ovarian carcinoma.
5-Azacitidine
Antimetabolite. Has a nitrogen on the 5 carbon where methylation typically occurs. Inhibition of methylation = inhibition of gene expression. 3rd nitrogen also makes it easier for the ring to split open and cause single stranded breaks when incorporated into the DNA where cytosine typically goes.
6–mercaptopurine/6-thioguanine
Antimetabolite. Purine analogue that stops purine biosynthesis.
Asparaginase
Enzyme. Cleaves asparagine to limit protein synthesis in leukemia cells. Decreased protein synthesis will have adverse affects on the liver and pancreas. Also since the enzyme comes from E. Coli you can have immune reactions against it.