Week 4- Inhalational Lung Disease Flashcards
(49 cards)
What are some origins of pollutants that can cause lung disease?
- natural (sand)
- combustion (exhaust, cigarettes)
- synthetic
- agriculture
- chemical reactions
- unknown
- indoor (cleaning products
- outdoors (ozone)
What are some possible patterns of exposure to lung pollutants?
- magnitude of exposure (insidious vs accidental?)
- context
- persistence (acute? chronic?)
- duration (steady? intermittent? diurnal?)
- geography
- conditions (exertion? PPE?)
What 3 factors determine the lung response to an inhaled pollutant?
- Deposition
- Clearance/Detoxification
- Biological activity of the substance
What determines the deposition of noxious gases and particles in the respiratory tract?
Gases
- depends on solubility (henry’s constant, e.g. gas:liquid ratio)
- higher gas:liquid deposits in the alveoli
- lower gas:liquid deposits in nasopharynx/upper resp (e.g. ammonia)
Particles
- depends mostly on particle size (sizes: thoracic, coarse, fine, ultrafine). Exhaust and cigarette smoke is ultrafine
What are the 3 patterns of deposition and what do they depend on? How could a pollutant be absorbed systemically?
Impaction (large airwyas, upper airways)
Sedimentation: (small airways)
Diffusion (alvoeli) ***may be able to enter the blood through this route and be absorbed systemically**
What are the lung’s natural defences?
- nasal filtering
- cough/sneeze reflex
- mucociliary escalator
- alveolar macrophages
- bactericidal enzymes
- lymphatic system
What determines whether a pollutant is cleared/detoxified from the lungs?
- the site of deposition
- the health of natural lung defences
What are some pathophysiologic mechanisms by which pollutants cause lung disease?
Non-specific chemical reactivity
- ROS
- strong acid or base
- inflammation–>necrosis–>fibrosis
Specific toxic effects
- DNA mutation
- invoke allergic response
- change O2 carrying capacity
What are some disease states that can caused (and exacerbated) by pollutant exposure?
Upper resp tract:
- rhinitis
- laryngitis
Upper airways
- bronchitis
- bronchiolitis
Small airways:
- asthma
- COPD
Diffuse:
- Cancer
- interstitial disease
What are some strategies to diagnose an occupational/environmental disease?
- acquire MSDS to know what you could be dealing with
- take a thorough history
- can send patient home with a spirometer and have them do PEF several times a day to establish whether the symptoms aer due to work exposure
How to minimize the incidence of diseases caused by environmental exposure?
Good industrial hygiene:
- Engineering controls
- administrative controls
- PPE
Government control
- regulations
- surveilllance for sentinel cases
Classification of lung malignancies
Small-cell carcinoma (15%)
Non small cell carcinoma (85%)
- adenocarcinoma
- squamous cell carcinoma
- large cell carcinoma
Mesotholioma
Pathologic characteristics of squamous cell carcinoma
- central
- often cavitate
- keratinization
- intracellular bridges
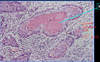
Pathologic features of adenocarcinoma
- 2/3 peripheral, 1/3 central
- gland formation +/- mucin production (PAS stain)

What is bronchoalveolar carcinoma? AKA?
AKA: adenocarcinoma in-situ (preferred name)
Tumour cells grow along the surface of the alveoli, BUT can have mixed AIS and invasive adenocarcinoma.

What are the pathologic features of small cell carcinoma?
- central
- small, dark cells with hyperchromatic nucleus (salt and pepper chromatin)

What are the pathologic features of large cell carcinoma?
- peripheral
- large polygonal cells, poorly differentiated

Why do we group lung cancers in small cell vs. non-small cell?
Within NSCLC, how and why do we group into squamous and non-squamous?
SCLC vs. NSCLC:
- Non-small cancers are treated primarly by surgical resection.
- Small cell cancers are more disseminated and are treated by chemotherapy
Squamous vs. Non-squamous
- subtype using TTF-1 and p63 expression
- no targeted therapy is available for squamous.
What targeted therapy is available for adenocarcinomas?
eGFR mutation positive –> tyrosine kinase inhibitor
ALK mutation positive –> crizotinib
**these are almost always mutually exclusive, and are not found in all patients**
What type of syndromes can local spread of lung cancer cause?
superior vena caval syndrome (facial swelling, cyanosis, protrusion of neck veins)
pancoast tumour (brachial plexus compression –> arm pain)
Horner syndrome (compression of cervical sympathetic chain –> unilateral ptosis, miosis, anhidrosis
pleural effusion
pericardial effusion
What is the usual lymph node spread of lung cancer?
intrapulmonary–?hilar nodes–>tracheobronchial nodes –>mediastinal nodes–>cervical, supraclavicular, axillary, distance nodes
What are the most common metastasis in lung cancer? How are they spread?
They are spread hematogenously
- adrenals (50%)
- liver (30-50%)
- brain (20%)
- bone (20%)
What are the clinical procedures used to acquire tissue to make a diagnosis of lung cancer?
- For central (squamous and small cell) can often do cytology on the sputum
- Bronchoscopy: lavage, brush, biopsy and do cytology/histology on each.
- Endobronchial ultrasound
- Trans-throacic fine needle aspirate
- Trans-thoracic core needle aspirate
Are primary or metastatic tumours of the lung or pleura more common?
Metastatic tumours are more common for both



