18. Atopy, allergy and dht 2 Flashcards
(23 cards)
Detection of allergen-specific IgE in vivo
Skin-prick testing
Allergen extract applied as drops
Top layers of epidermis punctured with lancet
A wheal with flare response after 15 minutes is positive
Result needs interpretation in clinical context
Detection of allergen-specific IgE in vitro
Performed by radioallergosorbant (RAST) assay a very long time ago
Now usually by ELISA, but term ‘RAST’ still widely used clinically
- Plastics coated with purified allergen of interest. Incubate with patient serum
- IgE antibodies in sera of sensitised patient bind to allergens
- Immobilised IgE antibodies detected with polyclonal anti-IgE detection antibody
Treatment of allergy
Pure symptom relievers (don’t act on mediators, but act on other pathways that oppose actions of mediators
- Nasal decongestants
- eg oxymetazoline
- Act on α1 adrenoreceptors to cause vasoconstriction
- Only for short-term use
- Topical and systemic
- B2 agonists
- Eg salbutamol
- Act on lung B2 adrenoreceptors, cause smooth muscle relaxation
- Epinephrine/adrenaline
* Systemic adrenergic effects oppose vasodilatation and bronchoconstriction
Treatment of allergy: drugs acting on early-phase mediators
Mast cell stabilisers act on mast cells
H1 antihistamines act on histamine
Leukotriene receptor antagonists act on leukotrienes

Mast cell stabilisers
Eg sodium cromoglycate
Reduce mast cell degranulation by unknown mechanism
Not orally absorbed – topical use only - e.g. eye drops
Short half-life requires frequent dosing
Main benefit is steroid-free, but efficacy very poor
H1 antihistamines
Inverse agonists at H1 histamine receptor
Best used before exposure to allergen
1st generation eg chlorpheniramine
- Considerable sedation, drug interactions
2nd generation eg cerizine, loratidine, desloratidine, fexofenadine
- No/ minimal sedation, once-daily
Leukotriene receptor antagonists
Only UK drug is montelukast
Effective in reducing early allergic responses, but inferior to H1 antihistamines
Unlike anti-histamines, beneficial in chronic asthma, which is the main indication for their use
Treatment of allergic disease: corticosteroids
Steroids reduce immune activation by altering gene expression in numerous cell types, including T cells, B cells and cells of the innate immune system.
Delayed onset of action and must be taken regularly

Corticosteroids
Inhaled
- Eg beclamathosome, fluticasone
Nasal
- Eg beclamathasone, mometasone, fluticasone
Also for skin (eg hydrocortisone) and ophthalmic drops
- Topical may cause local and even systemic side effects
Oral , intravenous and depot preparations available
Treatment of allergic disease: omalizumab
Omalizumab is a monoclonal antibody directed against IgE, used for atopic asthma
Binds Fc region of IgE to complex it in serum and limit free amount of it which can bind antigen

Allergen-specific immunotherapy
Allergen doses administered by subcutaneous injection or sublingually
Provide long-term protection
Mainly venom allergy and rhinitis
Multiple immunological effects:
- Induce regulatory T cell responses to allergens
- Reduce Th2 responses
- Induce allergen-specific IgG antibodies
- Reduction in mast cell responsiveness
- Reduce allergen-specific IgE levels
Type IV, delayed-type hypersensitivity
Mediated by antigen-specific effector T cells
- Antigen-specific: specific antigen stimulus needed, which is processed and presented to relevant T cells which carry out the reaction
- Effector T cell: T cells that have previously met antigen and are ‘primed’ to produce a rapid, robust response
Because it takes time to process and present antigen, these reactions do not develop for at least 24 hours following exposure
Example: contact dermatitis
Contact dermatitis: sensitisation
Sensitising agents are typically highly reactive small molecules which can penetrate skin
- These react with self proteins to create protein-hapten complexes that are picked up by Langerhans cells, which migrate to regional lymph nodes
- The Langerhans cells process and present the antigen together with MHCII
- In some susceptible individuals, the complexes are recognised as foreign
- The activated T cells then migrate to the dermis
Hapten = small molecule which cannot produce an immune response by itself, but can bind to a protein to alter its immunogenicity
Contact dermatitis: elicitation
Elicitation is when you meet antigen again after sensitisation
Chemokines recruit macrophages
- Th1 cells secrete IFN gamma: increases expression of vascular adhesion molecules, activates macrophages
- TNF alpha/ beta: local inflammation
Poison ivy
Pentadecacatechol is a poison ivy lipid that may cross the skin and modify intracellular proteins
These proteins are processed and presented with MHC1 to CD8 T cells which then cause contact dermatitis
Again, not everybody is susceptible
Native Americans would feed their babies poison ivy to generate tolerance
Patch test for contact dermatitis
Antigen-impregnated patch placed on back
Nickel, chrome, cobalt, epoxy resin, lanolin etc
Results read after 2 days (cw skin-prick testing which is immediate)
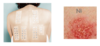
Difference between contact dermatitis and type 1 allergy


Skin prick testing vs patch testing


Tuberculin skin test (TST)
Used to determine EXPOSURE to TB
Chemoprophylaxis may be indicated to reduce risk of reactivation
Tuberculin injected intradermally (tuberculin=complex mixture of antigens derived from MTB)
Local inflammatory response evolves over 24-72 hours if previously exposed
Fairly poor test for active TB
Mechanism of TST
- Antigen is injected into subcutaenous tissue and processed by local antigen-presenting cells
- A TH1 effector cell recognises antigen and releases cytokines which cat on vascular endothelium
- Recruitment of phagocytes and plasma to site of antigen injection causes visible lesion
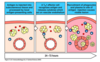
IGRA (interferon gamma release assay) to detect TB-specific Th1 cells
- MTB peptides added to blood in laboratory
- Antigen presenting cell presents peptide with MHC2 and secretes IL-12
With previous TB exposure:
- Effector memory Th1 cells recognise antigen. This is a secondary immune response so they are ‘primed’ and release cytokines within this short timeframe
With no previous TB exposure:
- No primed effect memory T cells specific for MTB. No interferon gamma produced in a short timeframe
IGRA: positive test by two methods
ELISPOT method (T-SPOT)
ELISA method (quantiferon gold)



