If you’ve ever walked out of an A-level Chemistry exam thinking, “I knew that… why didn’t I get the marks?”, there’s a good chance an “extended response” question was involved.
These questions look deceptively simple: “Explain how increasing temperature affects the rate of reaction.”
Easy, right?
And yet, these are some of the most commonly underperformed questions across A-level Chemistry exams, whether you’re studying under AQA, Edexcel, or OCR.
So what’s going on?
We’re going to answer that in this A-level Chemistry study guide, the first of five in Brainscape’s A-level Academy. We also have certified A-level Chemistry flashcards for the following boards, which you can start studying for free right away:
- AQA A-Level Chemistry flashcards
- OCR A-Level Chemistry flashcards
- Edexcel A-Level Chemistry flashcards
- OCR B / MEI A-Level Chemistry flashcards
- WJEC / Eduqas A-Level Chemistry flashcards
This is a powerful revision tool designed by our team of A-level educators to help you systematically learn and memorise the chemistry content you need to know to confidently answer the questions we’ll be talking about in this guide.
(They also include three free A-level Chemistry practice test papers.)
Why Extended Response “Explain” Questions Are So Tricky
Across all major exam boards, extended response “explain” questions are designed to test your ability to:
- Build a clear chain of reasoning
- Link ideas logically
- Use precise scientific language
- Avoid vague or incomplete statements
In other words, it’s not just about what you know (which is, of course, important) but how well you can connect it. It’s less like listing facts, and more like telling a tightly structured scientific story.
What A-Level Examiners Are Actually Looking For in Extended Response Questions
Marks are awarded for linked, coherent steps, not isolated points.
That means that one brilliant sentence won’t earn you the marks you’re after; and neither will listing several disconnected ideas.
Instead, you need to provide a logical chain where each idea builds on the previous one.
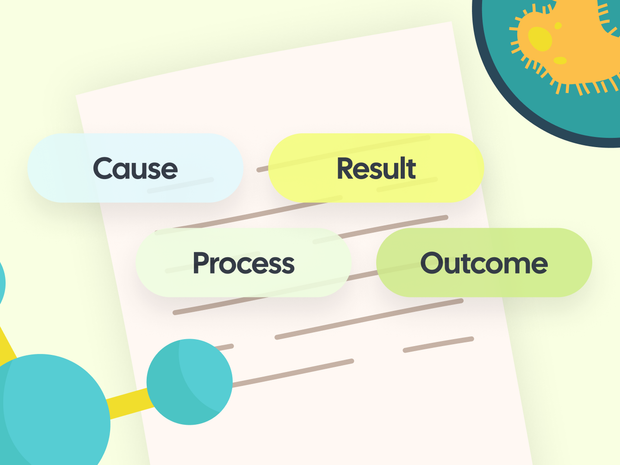
The “Chain of Reasoning” Model
A strong answer typically follows this pattern:
Cause → Process → Result → Outcome
Let’s look at that in action in a real extended response A-level Chemistry question.
Example question:
Explain how increasing temperature affects the rate of reaction. (4)
Weak answer (what many students write)
“Rate increases because particles have more energy and collide more.”
What’s wrong with this answer?
- It’s vague
- Missing key steps
- No mention of activation energy
- No clear chain
So, this might earn you 1–2 marks at best.
Remember, the mark allocation is a fundamental clue. You’ve got to earn those four marks!
Here’s how…
Strong answer (full-mark style)
“Increasing temperature increases the kinetic energy of particles. This means particles move faster and collide more frequently. More importantly, a greater proportion of particles have energy greater than the activation energy. Therefore, there are more successful collisions per second, increasing the rate of reaction.”
Now we’re talking! This answer:
- Provides a clear sequence
- Uses key terms precisely
- Has a logical progression
- Without any gaps in reasoning
All of this = happy examiner.
The Structure You Should Use Every Time
To make this kind of performance repeatable under exam pressure, use this simple framework:
Point → Because → Therefore
Or more explicitly:
- State what changes
- Explain why it changes
- Link to the outcome
Here’s another example so you can see this framework in action…
Question: Explain how changes in temperature and pressure affect the yield of ammonia in the Haber process. In your answer, refer to equilibrium position, rate of reaction, and the role of the catalyst. (10 marks)
Strong answer:
“The Haber process involves the reversible reaction:N₂(g) + 3H₂(g) ⇌ 2NH₃(g), which is exothermic.
Increasing pressure shifts the equilibrium to the right, producing more ammonia. This is because there are fewer moles of gas on the product side (2 moles) compared to the reactant side (4 moles), so the system responds by reducing pressure.
Increasing temperature shifts the equilibrium to the left, reducing ammonia yield. This is because the forward reaction is exothermic, so adding heat favours the endothermic reverse reaction. However, higher temperatures increase the rate of reaction, allowing equilibrium to be reached more quickly.
In industry, a compromise temperature (around 450°C) is used to balance a reasonable rate with acceptable yield, while high pressure (around 200 atm) is used to maximise ammonia production.
An iron catalyst is also used to increase the rate of both forward and reverse reactions equally, allowing equilibrium to be reached faster. However, it does not change the position of equilibrium or the overall yield.”
The Most Common Extended Response Question Mistakes (& How to Fix Them)
Let’s save you from the classic pitfalls.
1. Being Too Vague
“Particles have more energy” versus“Particles have greater kinetic energy, so more have energy above the activation energy” ✓
Always be specific.
And remember to pay attention to the command words used in the question.
Read: ‘How to Decode A-Level Chemistry Command Words’.
2. Missing the Key Concept
Every question is built around a central idea. For example:
- Rates → activation energy and successful collisions
- Bonding → intermolecular forces or structure
- Redox → electron transfer
- Equilibrium → position of equilibrium and Le Chatelier’s principle
If you miss that core idea, your answer becomes vague or incomplete, even if the rest of your explanation is well written.
So, before you start writing, ask yourself: “What is the main concept this question is really testing?” Then, make sure that concept is clearly stated, accurately explained, and explicitly linked to the final outcome.
Speaking of which…
3. Not Linking Ideas
“Particles move faster. There are more collisions.” “Particles move faster, so they collide more frequently” ✓
Use linking words and phrases like “so”, “therefore” and “which means”.
4. Writing in Bullet Points
Most exam boards reward continuous logical explanation, not fragmented notes. So, while it might be tempting to write out answers in bullet points (maybe to save time) it’s risky.
5. Stopping Too Early
Students often write a couple of steps… and then stop, thinking that they’ve satisfied the question. But if the question counts for (let’s say) six marks, examiners are usually looking for 4-6 linked ideas! Make sure you deliver that before moving on to the next question.
A Quick Checklist Before You Move On
When you finish an A-level Chemistry extended response question, mentally check:
- Have I used key scientific terms?
- Have I linked each step clearly?
- Have I explained why, not just what?
- Have I taken the idea all the way to the final outcome?
If yes, you’re in strong territory. Try to memorise this checklist so that you can apply it during the examination to prevent yourself from leaving precious marks on the table.
What’s the Best Way to Practise Extended Response Questions Effectively?
The honest truth is that you don’t get better at these by rereading your notes.
You get better by:
- Practising exam-style questions
- Comparing your answers to mark schemes
- Training yourself to think in chains, not fragments
Step 1: Practise With Real Exam-Style Questions
Work through extended response questions across multiple topics.
(We’ve created full A-level Chemistry practice papers to help with exactly this and you’ll get these papers for free when you download Brainscape’s A-level Chemistry flashcards.)
Step 2: Analyse Mark Schemes
Don’t just check to see if you got the answer right or wrong.
Scrutinize the structure, wording, and linking. Did you provide a fact for every mark allocated? Did you logically connect them and provide reasoning? The more you identify these discrete steps in answering A-level Chemistry extended response questions, the more natural and automatic they’ll become.
Step 3: Train Your Brain With A-level Chemistry Flashcards
In order to successfully answer questions like these, you need strong content knowledge and the ability to easily retrieve that knowledge from scratch.
This is where a digital flashcard app like Brainscape comes in.
We’ve worked with a team of A-level educators and Chemistry professors to distill the complete syllabi for dozens of A-level boards into collections of adaptive digital flashcards. (Our expert-vetted and certified collections have little green check marks next to them. The rest have been made by other learners in Brainscape.)
You can study these cards anytime, anywhere, on any device with an in-built spaced repetition algorithm showing you exactly the concepts you need to see, when you need to see them, to lock them in for the long term!
Final Takeaway for A-level Chemistry Students (& Educators)
Extended response questions fundamentally boil down to connecting what you already know clearly, logically, and completely.
Once you master that, these questions go from intimidating to some of the most reliable marks on the paper.
And that’s exactly where you want to be on exam day!
Additional A-level Chemistry Guides & Resources:
- How to Avoid Losing Marks on A-Level Chemistry Calculation Questions
- Common A-Level Chemistry Mistakes To Avoid
- How to Decode A-Level Chemistry Command Words
- How To Score Full Marks On A-Level Chemistry Practicals
- Brainscape Periodic Table Flashcards
- Free Revision Planner Templates & Exam Countdown Sheets