ENDO Portion Flashcards
(135 cards)
what is the most common cause of hyperthryoidism?
GRAVES DISEASE!!!
hyperthyroidism:
Graves disease
what is important to knwo about this?
who is it common in?
what causes this?
3 things it leads to?
MOST COMMON CAUSE OF HYPERTHYROIDISM
women more common 20-40 years old!!
autoimmune TSH-R AB-IgG antibodies aka TSI directed to TSH receptor over-activate gland leading to hypersecretion
leads to: hypertrophy, hyperplasia, commonly goiter!!

hyperthyroidism:
graves disease
13 sxs
2 3 key ones you need to remember
- hyperactivity, irritability, restlessness
2. heat intolerance, sweating
- palpatations
- increased appetite, weight loss
- tachycardia
- arrythmia
- fine tremor
8. goiter
- warm, oily hair
- proximal muscle weakness
- opthalmopathy
-proptosis/exopthamos
-lid lag
-
dermopathy
- pretibial myxedema - hyperreflexes

hyperthyroidism:
Graves disease
OPTHALMOPATHY
3 things causes?
why?
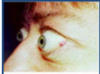
unique to graves disease

- proptosis/exopthalmos “lid lag”
- conjunctival inflammation/edema
- corneal drying
******occurs because the lympocytes infiltrate the orbit, muscles, eyelids and may cause diplopria and compression of optic nerve*****
hyperthyroidism:
Graves Disease
DERMOPATHY
what is the name for this?
what does this cause?
apperance?
3% occurance
“pre-tibial myxedema”
*******noninflammatory induration and plaque formation of the pre-tibial area leading to thickened skin, and orange skin appereance*****
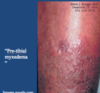
hyperthyroidism:
Graves disease
2 tests to check
what don’t you need to check?
- very low TSH
since the T3 and T4 high, negative causes this to be low
2. total and T4 elevated
**don’t need to check TSH-R AB for dx**
hyperthyroidism:
Graves disease
9 tx options?
what is the toc?
who do you use each in?
- endocrinology consult essential
- propanolol for sxs
-
methimazole (thiourea) -MILD TO MOD 12-18 months!
- inhibits thyroid peroxidases and block organification of iodine to decrease hormone production
-monitor WHB, pruirits, and FT4
- propylithiouricil (PTU) (thiourea) if pregnant
- saturated iodine solution- severe
- iodinated contrast agens- severe or thyroid storm
- prevents conversion of T4 to T3 - glucocorticoids-severe
- prevents conversion -
radioative iodine-DEFINITIVE TX and TREATMENT OF CHOICE IN THE US!!
- destroys the overactive gland because concentrates here
- uses I-131 - thyroidectomy
no longer TOC but used in children or pregnancy or those that can’ be controlled with medication
hyperthyroidism:
Graves disease
what do you need to keep in mind about the toc for this? what is it? what might worsen?
radioactive iodine
used to destroy the gland because it concentrates here
permanent hypothyroidism often develops within 1 year and may need replacement tx FOR LIFE!!!!
***opthalmopathy may worsen esp in smokers with this tx***
hypothyroidism:
Hashimoto’s thyroiditis
what is important to remember about this?
what type of disease?
how does it show up?
2 things ath cause this
what to keep in mind?
MOST COMMON CAUSE OF HYPOTHYROIDISM
AUTOIMMUNE, with insidious onset
thyroperoxidase and thyroglobulin antibodies present in high titers
*****keep in mind, this can itially cause a hyperthyroidism from release of stored hormone, but the end result is hypothyroisim*****

hypothyroidism:
Hashimoto’s thyroiditis
5 early sxs
8 late sxs
early:
- cold intolerance
- dry hair, hair loss
- headaches
- mennorhagia
- thin, brittle nails
late:
- slow speech
2. hoarse voice
3. weight gain
3. goiter
4. alopecia
4. facial and eyelip puffiness
5. bradycardia
6. edema non pitting
7. myxedema
8. pleural/pericardial effusion

hyperthyroidism:
Graves disease
in txing it with thiourea drugs what percent will have reccurance?
(propylithiouricil or methimazole)
50%
reccurence are common if tx is only with thiourea, propylthiouracil or methimazole
what are 4 complications that can come from graves disease?
- CV
- ocular
- psychological complications
- post tx hypothyroidism common, but easily txed
thyroid storm
what is this?
3 labs?
4 sxs?
3 tx options?
rare, but life-threatening of extreme hyperthyroidism, can be fatal and need to be admitted and txed STAT
elevated T3 and T4
decreased TSH
high fever
tachycardia
sweating
delierum
tx:
- satruated iodine solution
- iodinated contrast agents
- glucocorticoids
what are the 5 causes of hypothyroidism?
- hasimotos thyroiditis
2. congenital hypothyroidism
- idiopathic
- iatrogenic
- drug induced- amiodarone
hypothyroidism:
congenital
what testing?
why is it key to identify?
screening of TSH in newborns should elimate disease
early detection is key to prevent cretinism** and **hypodevelopment

hypothyroidism:
hashimotos thyroiditis
myxedema
what can this lead to?
5 sxs?
abnormal interstitial fluid acculuation in skin giveing it a waxy/coarsened (non-pitting) appearence
can lead to mydxedema crisis EMERGENCY:
severe form of hypothyroidism
- bradycardia
- CNS depression (coma)
- respiratory depression
- hypotension
- hypothermia
hypothyroidism:
hashimotos thyroiditis
2 complications
4
1
-
Cardiovascular
- pericardial effusions
- cardiomyopathy
- accerlerated CHD
- HF - encephalopathy
coma/confusion
hypothyroidism:
Hashimotos thyroiditis
5 dx?
- High TSH
- low T3/T4
- increased triglycerides
- decreased HDL
- anti-thyroid antibodies
hypothyroidism:
hashimoto’s thyroiditis
what is the tx?
how is it dosed?
monitoring?
goal? time frame?
levothyroxine (synthetic T4)
a. start 50-100 ug/day and titrate to full dose over time, following TSH levels
b. recheck TSH levels every 2-3 months until normal
GOAL: clinically euthyroid state (normal)
**sxs improve slowly over months**
hypothyroidism:
iatrogenic (we caused it)
tx?
radioative iodine for initial hyperthyroidism that leads to gland destruction
thyroiditis:
suppurative
what is this caused by?
3 sxs?
1 dx?
1 tx?
caused by gram positive bacteria
STAPH AUREUS
1. tender thyroid
- fever
- pharyngitis
DX:
fine needle aspiration
Tx:
drainage
thyroiditis:
De quervains aka granulomatous
what is this?
when is it most common?
when does it occur?
explain the progression?
2 dx?
- tx?
MOST COMMON CAUSE OF PAINFUL THYROID GLAND
***peaks in the summer….weird****
MC POST VRAL INFECTION
thyrotoxicosis initially presents followed by hypothyroidism and euthyroid within 12 months
DX:
- markedly increase ESR
- very low anti-thyroid bodies
Tx:
TOC=aspirin!!!
bb
ionated constrast agent
thyroiditis:
drug induced amiodarone
what does the medication contain?
what percent get this?
3 progression steps?
contains 37% iodine by weight or 75 mg per tablet
causes thyroid dysregulation in 20% of patients
Progression:
- can cause rise in T4 during first month of tx
- causes cellular resitsance to T4
- hypothyroid picture ensues with elevated TSH
thyroiditis:
fibrous thryoiditis (riedel)
who is this in?
what hapens to the thyroid?
key description
what happens to RAI?
ab?
1 dx method
1 tx method?
rarest form of thyroiditis, 80% are in females
formation of dense fibous tissue in the thyroid
causes
hard “woody” asymmetric thyroid feeling
the fibrosis can spread outside of the thyroid
radioactive iodine is decreased in involved areas of the thyroid
antibodies may be present in 45%
DX:
BIOPSY
tx:
tomoxifan