Chapter 15 Flashcards
(41 cards)
What are fatty acids?
long chain carboxylic acids
carboxyl carbon=number 1
carbon number 2=alpha carbon
saturated fatty acids have no double bonds
palmitate: primary end product of FA synthesis (C16:0)
What are unsaturated fatty acids?
one or more double bonds
can synthesize a few; most come from essential fatty acids in diet
–transported as triglycerides from intestine in chylomicrons
–ex. inolenic acid and linoleic acid (important membrane phospholipids to maintain nomral fluididty of cell membrane)
omega: position of last double bond relative to end of chain
arachidonic acid: precursor for prostaglandins, thromboxanes, leukotrienes
double bonds in FA are in cis configuration
trans double bonds: unnatural , margarine; decrease membrane fluidity; Increase risk of atherosclerosis (and saturated fatty acids)
Waht is the cardioprotective effects of omega 3 acids?
decreased risk of cardiovascular disease
replace some of the arachidonic acid (omega 6 fatty acid) in platelet membranes and lower production of thromboxane and the tendency of platelets to aggregate
–decrease serum triglyerides
found in cold water fish, nuts and seeds
How are lipids digested?
high fat meals contain gram level amounts of triglycerides and milligram amounts of cholesterol and cholesterol esters
–intestinal lumen: bile is secreted by liver to emusify lipids
–pancreas: secretes pancreatic lipase, colipase, and cholesterol esterase that degrade lipids to 2 monoglycerides, fatty acids and cholesterol
–lipids absorbed and reesterfied to triglycerides and cholesterol esters
–packages with apoprotein B48 and other lipids into chylomicrons
–litle lipid loss in stools
–defect=steatorrhea (excessive amounts of lipids in stool)
How are fatty acids synthesized?
excess dietary glucose is converted to fatty acids in liver and then sent to adipose tissue for storage
–adipose tissue syntehsizes small quantities of fatty acids
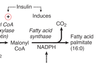
What affect does insulin have in converting glucose to acetyl CoA in liver?
promotes many steps:
–glucokinase: induced
–PFK-2/PFK-1: PFK2 dephosphorylated
–pyruvate dehydrogenase: dephosphorylated
What enzymes of fatty acid synthesis are affected by insulin?
–acetyl CoA carboxylase: dephosphorylated, activated
–fatty acid synthase: induced
What is the citrate shuttle?
transports acetyl CoA from mitochondria to the cytoplasm for fatty acid syntehsis
acetyl CoA combines with OAA in mitochondria to form citrate
citrate transported into cytoplasm
citrate lyase forms acetyl Coa and OAA from citrate
OAA returns to mitochondria
reaction: additional source of cytoplasmic NADPH in liver and adipose tissue, supplementing that from the HMP shunt
activated by: insulin and high energy status

What is the function of Acetyl CoA carboxylase?
acetyl CoA is activated in cytoplasm for incorporation into fatty acids by acetyl CoA carboxylase
–rate limiting enzyme of FA synthesis
requires biotin, ATP, and CO2
activated by insulin (dephosphorylated) and citrate
CO2 added to form malonyl CoA isn’t incorporated into FA–removed by FA synthase during addition of acetyl group to fatty acid

What is the function of fatty acid synthase?
pallmitate synthase
palmitate is the only fatty acid that human can make de novo
cytoplasm
induce in liver after a meal by high carboxydrate and inulin
contains acyl carrier protein–requires vitamin pantothenic acid
–malonyl CoA is substrate used by FA synthase–only carbons from acetyl CoA portion are incorporated into FA
–FA is derived entirely from acetyl CoA
NADPH is required
–8 acetyl CoA required to make palmitate
Can fatty acyl CoA be elongated and desaturated?
yes by enzymes in SER
cytochrome b5 can desaturate
–can’t introduce double bonds past position 9 in fatty acid
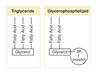
What are triglycerides?
sotrage form of fatty acids
3 fatty acids (fatty acyl CoA) to glycerol
formed in iver and adipose tissue
liver sends triglycerides to adipose tissue packaged as VLDL
small amount of triglyceride may be stored in liver
What are the sources of glycerol 3 P for triglyceride synthesis?
- reduction of dihydroxyacetone pohsphate (DHAP) from glycolysis by glycerol 3-P dehydrogenase (enzyme in adipose tissue and liver)
–adipose tissue dependent on glucose uptake via GLUT 4 transport (stimulated by insulin ensuring good supply of DHAP for TG synthesis)
- phosphorylation of free glycerol by glycerol kinase (enzyme only in liver)
–allows liver to recycle glycerol released during VLDL metabolism (insulin) back into new triglyceride syntehssis
–during fasting (glucagon), enzyme allows liver to trap glycerol released into blood from lipolysis in adipose tissue for later conversion to glucose

What are glycerophospholipids?
used for membrane syntehsis and producing hydrophilic surface layer on lipoproteins like VLDL
–reservoir of second messengers such as diacylglycerol, inositol 1,4,5, triphosphate and arachidonic acid
structure is similar to TG except last fatty acid is raplaced by phosphate and water soluble group like choline or inositol
What are lipoproteins?
tiglycerides and cholesterol are trnasported in blood as LP
desnity increases with percentage of protein
least to most dense:
chylomicrons < VLDL < IDL < LDL < HDL

What is a cholesterol ester?

What arethe steps of lipoprotein metabolism?

What are chylomicrons?
primarily triglyceride particles and small quanity of cholesterol esters
transport dietary triglyceride to adipose tissue and muscle
have apoC-II, apoE, apoB, apoB-48
assembled from dietary TG–mainly longer chain fatty acids, including essential FA–cholesterol esters and 4 lipid soluble vitamins
core lipid is surrounded by phospholipids (increase solubility of chylomicrons in lymph and blood)
ApoB48: required for release from epithelial cells into lymphatics
–leave lymph where thoracic duct joints left subclavian vein so bypass liver
after high fat meal, chylomicrons cause serum to become turbid or milky
in blood, chylomicrons acquire apoC-II and apoE from HDL particles
in capillaries of muscle and adipose tissue, apoC-II activates lipoprotein lipase, FA released enter tissue for storage and glycerol is retrieved by liver which has glycerol kinase
remnant is picked up by hepatocytes by apoE receptor
–dietary cholesterol and remaining TG is released in hepatocyte

What is the function of ilpoprotein lipase?
required for metabolism of chylomicrons and VLDL
induced by insulin
tranpsorted to the luminal surface of capillary endothelium so in direct contact with blood
hydrolyzes FA fro mTG carried by chylomicrons and BLDL
activated by apoC-II
What are very low density lipoproteins (VLDL)?
primarily triglyceride particles and small quantities of cholesterol esters
transport TG (containing FA newly syntehsized from excess glucose or retrieved from mchylomicron remnants) synthesized in liver to adipose tissue and muscle
have apoC-II, apoE, apoB and apoB-100
apoB111 is added in the hepatocytes to mediate release into blood
VLDL acquire apoCII and apoE from HDL in blood
metabolized by lipoprotein lipase in adipose tissue and muscle

What are the steps of chylomicron and VLDL metabolism?

What are VLDL remnants?
intermediate density lipoproteins (IDL)
after TG is removed from VLDL
portion of IDL is picked up by hepatocytesthrough apoE receptor but some IDL stays in blood where they are further metabolized
transition particles between TG and cholesterol transport
can acquire cholestreol esters ransferred from HDL particles and become LDLs

What is a low density lipoprotein (LDL)?
primarily cholesterol particle
most cholesterol measured inblood is associated with it
deliver cholesterol to tissues for biosyntehsis
cholesterol is required for membrane synthesis, bile acids and salts in liver, steroid synthesis
80% of LDL are picked up by hepatocytes, rest are in peripheral tissue
apoB100 is only apoprotein
endocytosis of LDL is medaited by apoB 100 receptors (LDLD receptors) clustered in areas of cell membranes lined with protein clathrin