What is haemoglobin and what does it do?
- Haemoglobin is a tetrameric haem protein found in blood which carries oxygen from the respiratory organs to body tissues
- There, it releases the oxygen to permit aerobic respiration in order provide energy for metabolism
What is myoglobin and what does it do?
- Myoglobin is a monomeric haem protein found mainly in muscle tissue where it serves as an intracellular storage site for oxygen
- During periods of oxygen deprivation oxymyoglobin releases its bound oxygen which is then used for metabolic purposes
Describe the binding of oxygen to haemoglobin
- O2 binds to haemoglobin via the Fe atom in the haem prosthetic group
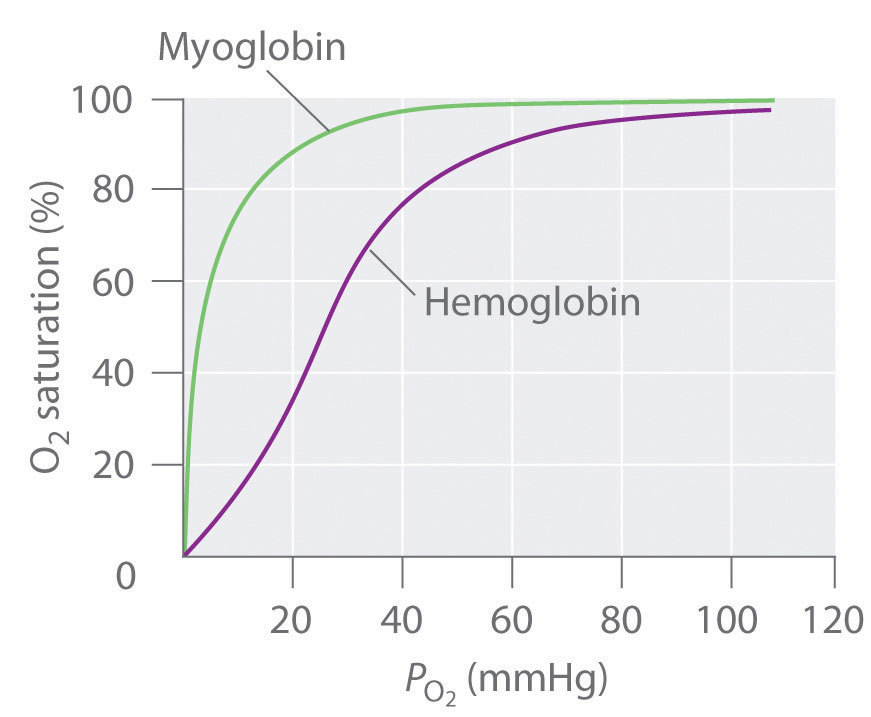
- O2 binding to haemoglobin shows a sigmoid shaped dependence on [O2]
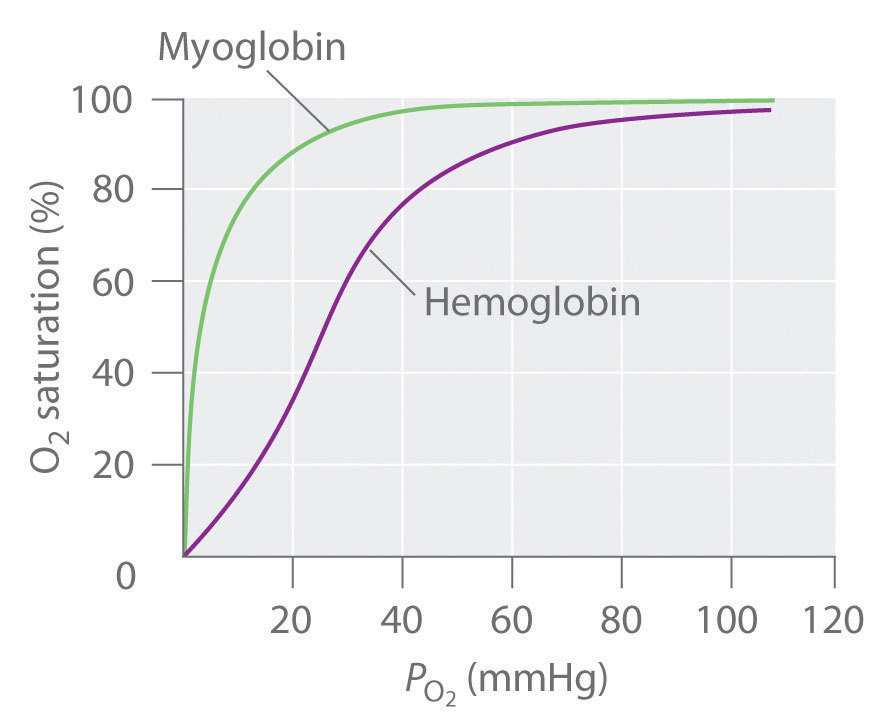
Describe the binding of oxygen to myoglobin
- O2 binds to myoglobin via the Fe atom in the haem prosthetic group
- O2 binding to myoglobin shows a hyperbolic dependence on [O2]
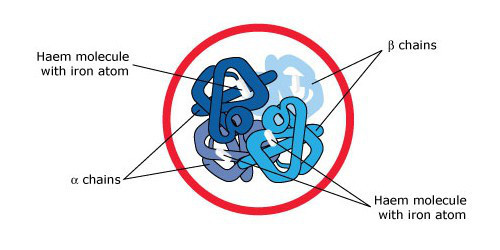
Describe the overall structure of haemoglobin
- Composed of 4 polypeptide chains in α2β2 tetramer
- Each chain contains an essential haem prosthetic group
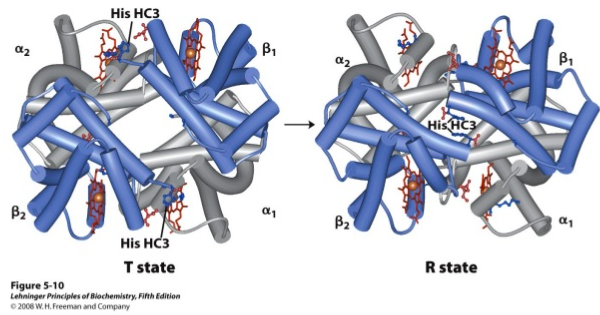
Explain how haemoglobin undergoes a structural change on oxygen binding
- Deoxyhaemoglobin exists in either the low affinity T state or the high affinity R state
- Oxygen binding promotes stabilisation of the R state
Why is the oxygen-dissociation curve for haemoglobin sigmoidal?
- Haemoglobin ‘co-operatively binds’ to oxygen
- The binding of one oxygen molecule promotes the binding of the next
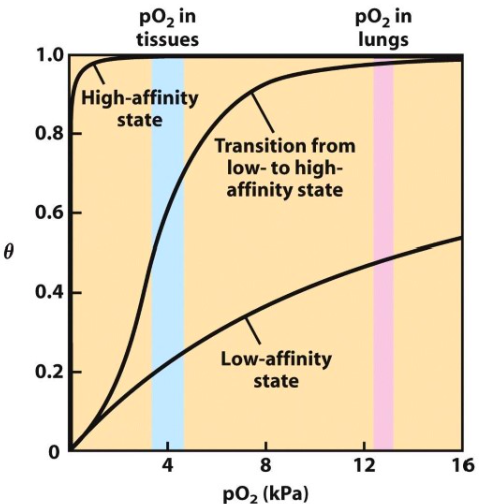
What is the benefit of the sigmoidal binding curve of haemoglobin?
- O2 can be efficiently transported from the lungs to the tissues
- Haemoglobin is more sensitive to small differences in [O2]
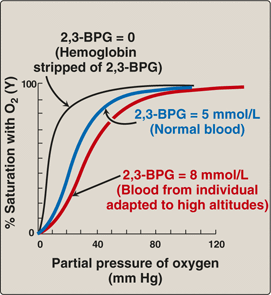
2,3-BPG is a molecule which regulates oxygen binding.
What is the effect of the binding of 2,3-BPG to deoxyhaemoglobin?
- 1 BPG binds per haemoglobin tetramer and decreases the affinity for O2
- [BPG] increases at high altitudes, promoting O2 release at the tissues
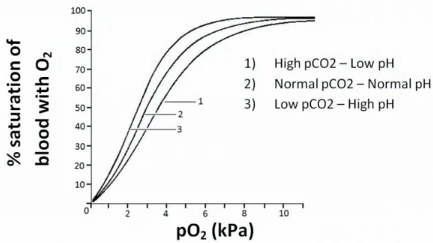
Describe he regulation of oxygen binding in terms of CO2 and H+
Binding of H+ and CO2 lowers the affinity of haemoglobin for oxygen
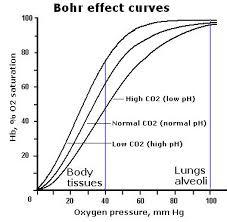
What is the benefit of the Bohr effect?
- Metabolically active tissues produce large amounts of H+ and CO2
- The Bohr effect ensures the delivery of O2 is coupled to demand
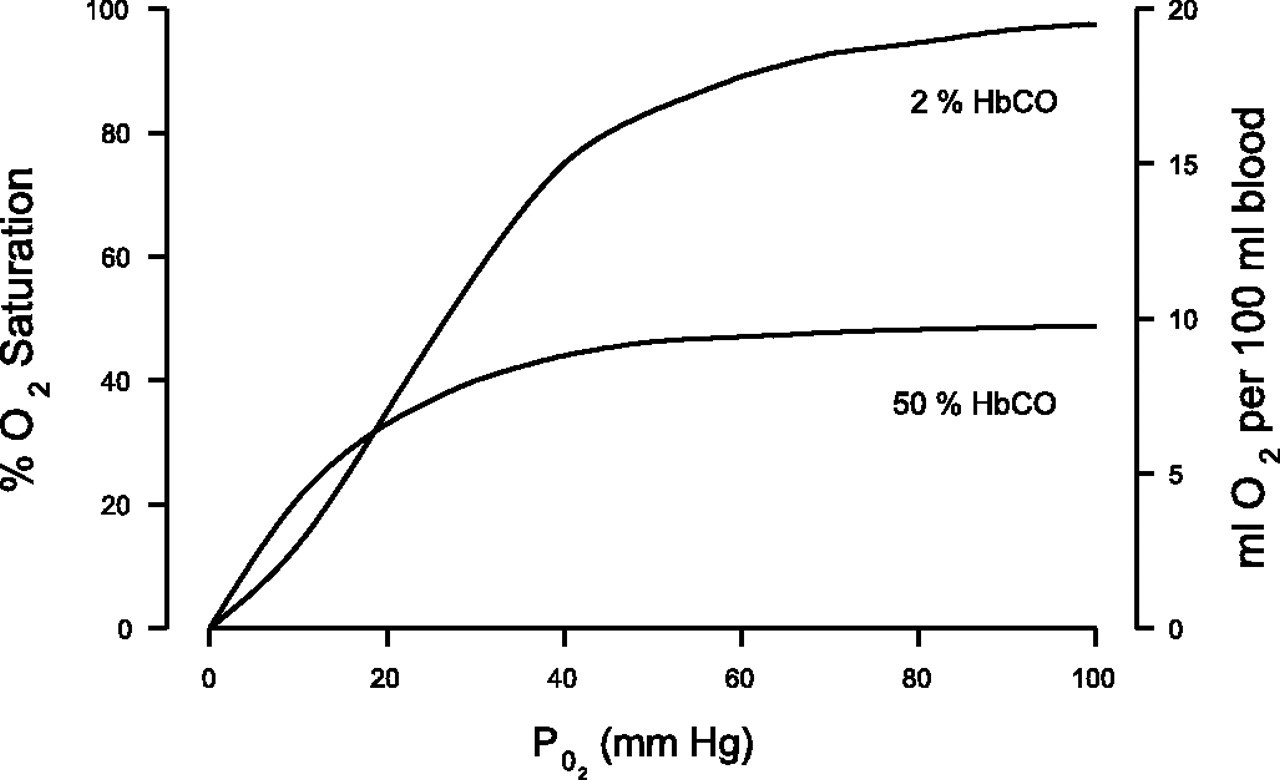
In terms of oxygen binding, explain why is carbon monoxide poisonous?
Carbon monoxide (CO) is a poison because it combines with ferromyoglobin and ferrohaemoglobin to block oxygen transport
CO binds to haemoglobin 250x more readily than O2.
Illustrate the impact of this on the oxygen binding curve of haemoglobin
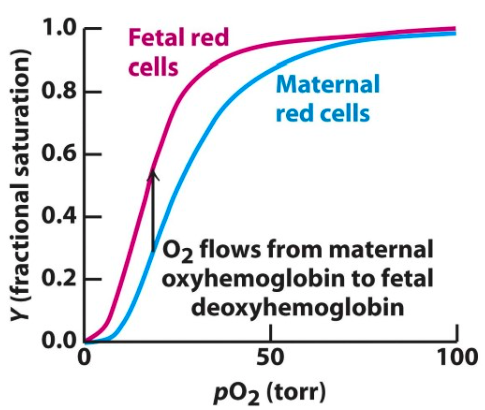
HbF is the major haemoglobin in foetal blood.
Why is it important?
Higher binding affinity of HbF for O2 than HbA allows transfer of O2 to foetal blood supply from the mother
Identify 2 properties of red blood cells with the HbS protein as seen in sickle cell anaemia
- More prone to lyse (anaemia)
- More rigid (black microvasculature)


